What is the latest evidence about the existence of long-COVID or post-COVID and its persistence for COVID-19 survivors? What evidence is currently available on the management of patients who have post viral fatigue syndrome due to COVID-19?
Download summary of evidence (pdf)
The following information resources have been selected by the National Health Library and Knowledge Service Evidence Virtual Team in response to your question. The resources are listed in our estimated order of relevance to practicing healthcare professionals confronted with this scenario in an Irish context. In respect of the evolving global situation and rapidly changing evidence base, it is advised to use hyperlinked sources in this document to ensure that the information you are disseminating to the public or applying in clinical practice is the most current, valid and accurate. For further information on the methodology used in the compilation of this document – including a complete list of sources consulted – please see our National Health Library and Knowledge Service Summary of Evidence Protocol.
Main Points
- Two distinct clinical scenarios are emerging: 1. patients with initially severe illness requiring hospitalization whose rehabilitation and recovery can be complex and slow; and 2. ongoing debilitating symptoms in patients who may not have required hospital admission initially. It is the latter who are sometimes referred to as ‘long COVID’ patients. ‘Long COVID’ is a term first coined on social media.
- There is an absence of higher-quality evidence as yet about the longer-term effects of COVID-19, but a growing body of anecdotal/observational evidence and some preliminary studies suggest persistence of at least one and sometimes several symptoms.
- Fatigue, dyspnea, breathlessness and cough, joint and muscle pain, chest pain and palpitations are among the most commonly reported symptoms.
- It is speculated that the virus may injure multiple organs and/or bodily systems. Potential long-term effects include damage to lungs, the immune system, the heart, and the brain, resulting in neurological complications, olfactory dysfunction and/or chronic fatigue similar to CFS/ME.
- There is potential for a significant and persistent negative mental health impact, based on previous experience with other pandemics.
- Many patients recover spontaneously (if slowly) with holistic support, rest, symptomatic treatment, and a gradual increase in activity. Indications for specialist assessment include clinical concern along with respiratory, cardiac or neurological symptoms that are new, persistent, or progressive.
- There are several ongoing studies investigating the aetiology, outcomes and management of persisting symptoms that will in time provide higher quality evidence.
Summary of Evidence
Two distinct clinical scenarios are emerging: 1. patients with initially severe illness requiring hospitalization whose rehabilitation and recovery can be complex and slow; and 2. ongoing debilitating symptoms in patients who may not have required hospital admission initially. It is the latter who are sometimes referred to as ‘long COVID’ patients. ‘Long COVID’ is a term first coined on social media.
COVID-19 can result in prolonged illness and persistent symptoms, even in young adults and persons with no underlying medical conditions who were not hospitalized1, 6. In a study by Carfi et al5 among patients who had recovered from COVID-19 at a Rome hospital, 87.4% reported persistence of at least one symptom, most often fatigue, dyspnea, joint pain, chest pain and cough. According to Greenhalgh et al7, approximately 10% of people experience prolonged illness after COVID-19.
Breathlessness, chest heaviness, muscle pain, palpitations and fatigue are among the continuing and debilitating symptoms being reported by people with COVID-19 often months after the onset of the disease and after they have been declared recovered10. Mardani12 states that longer-term complications may include heart failure, neurological disease and lung disease. Symptoms may include fatigue and brain fog, which may be related to cytokines that cross the blood-brain-barrier and affect the brain12, 23; and may be indicative of a post-viral syndrome associated with COVID-19. Insomnia, general myalgia, dermatological manifestations, exercise intolerance, temperature dysregulation and increased anxiety are other common symptoms12, 13, 14.
In a purposive sample of 100 survivors assessed 4 to 8 weeks after discharge from hospital, Halpin et al14 found that new illness-related fatigue was the most common reported symptom by 72% of participants who had required treatment in an intensive care unit and 60.3% managed in hospital wards without needing ICU care. The next most common symptoms were breathlessness (65.6% in ICU group and 42.6% in ward group) and psychological distress (46.9% in ICU group and 23.5% in ward group).
Marshall16 and Kemp35 suggest that the virus may injure multiple organs; and although the most severe infections also cause the worst long-term impacts, even mild cases can have life-changing effects ¾ notably a lingering malaise similar to chronic fatigue syndrome. Potential long-term effects include damage to the lungs, to the immune system and the heart, and to the brain, resulting in neurological complications such as delirium, confusion and memory loss16.
Olfactory dysfunction is another lingering symptom with up to 10% of patients reporting persistent loss or distortion of smell or taste eight weeks after symptom onset27, 32, 42. Chary et al29, however, conclude that although olfactory and gustatory dysfunctions related to COVID-19 are frequently reported and prevalent in mild symptomatic forms of the disease, recovery in most cases seems rapid and complete.
Carvalho-Schneider et al28 recommend a prolonged medical follow-up of patients with COVID-19 regardless of the severity of initial clinical presentation.
One of the most insidious long-term effects of COVID-19 is its least understood: severe fatigue16, 18. Over the past nine months, an increasing number of people have reported crippling exhaustion and malaise after having the virus. Symptoms such as foggy thoughts, breathlessness and exhaustion resemble chronic fatigue syndrome, also known as myalgic encephalomyelitis (CFS/ME); comparisons with CFS/ME are frequent in the literature8, 10, 12, 16, 17, 23, 45.
Pero et al9 speculate that psychiatric illness and fatigue may also be a long-term effect of COVID-19, as it was previously for the SARS epidemic. Lyons et al17 also caution about the potential for a significant and persistent negative mental health impact, based on previous experience with other pandemics.
Rooney et al45 conclude that physical function and fitness are impaired following SARS-CoV infection, and impairments may persist up to 2 years post-infection. However, there are concerns related to the impact of graded exercise therapy for managing post-viral fatigue in patients recovering from COVID-19, with NICE acknowledging that graded exercise therapy may not be appropriate for some patients46.
There are no definitive, evidence-based recommendations for the management of post-acute COVID-19 as yet; therefore, patients should be managed pragmatically and symptomatically: eg antipyretic for fever, breathing techniques for chronic cough, home pulse oximetry for monitoring breathlessness, pulmonary rehabilitation, staged return to exercise3. Greengalgh et al7 state that patients recover spontaneously (if slowly) with holistic support, rest, symptomatic treatment and gradual increase in activity. Indications for specialist assessment include clinical concern along with respiratory, cardiac or neurological symptoms that are new, persistent, or progressive.
There are several ongoing studies investigating the aetiology, outcomes and management of persisting symptoms that will in time provide higher quality evidence.
Irish and/or International Guidance
What does the World Health Organization say?
World Health Organisation (September 2020) What we know about long-term effects of COVID-19. The latest on the COVID-19 global situation and long-term sequelae[1]
COVID-19 can result in prolonged illness and persistent symptoms, even in young adults and persons with no underlying medical conditions who were not hospitalized. Little is known about the clinical course of COVID-19 following milder illness. Much is still unknown about how COVID-19 affects people over time. More time and research is needed to understand the long-term effects of COVID-19, why symptoms persist or recur, how these health problems affect patients, the clinical course and likelihood of full recovery.
This leaflet provides basic exercises and advice for adults who have been admitted to hospital with COVID-19, providing information on: managing breathlessness; exercising after leaving hospital; managing voice problems; eating and swallowing; managing attention and memory deficits; managing stress; and getting back to functional activities.
What does NICE say?
Includes recommendations on identifying people with ongoing symptomatic COVID-19 or post-COVID-19 syndrome; assessing people with new or ongoing symptoms after acute COVID-19; investigations and referral; planning care; management; follow-up and monitoring; sharing information and continuity of care; service organisation.
Point-of-Care Tools
What does BMJ Best Practice say?
BMJ Best Practice (2020) Coronavirus disease 2019 (COVID-19)[4]
There are no definitive, evidence-based recommendations for the management of post-acute COVID-19 as yet; therefore, patients should be managed pragmatically and symptomatically: eg antipyretic for fever, breathing techniques for chronic cough, home pulse oximetry for monitoring breathlessness, pulmonary rehabilitation, staged return to exercise. Many patients recover spontaneously with holistic support, rest, symptomatic treatment, and a gradual increase in activity.
What does UpToDate say?
UpToDate (2020) Coronavirus Disease 2019 (COVID-19)[5]
See Section: Recovery and Long-Term Sequelae
Systematic evaluation of the long-term sequelae of COVID-19 is lacking, but emerging data and evidence from other coronaviruses suggest the potential for ongoing respiratory impairment. Moreover, cardiac imaging studies have suggested the potential for cardiac sequelae after COVID-19, even among patients managed in the outpatient setting. Patients who were critically ill with COVID-19 may also be at risk for post-ICU syndrome [persistent impairments in cognition, mental health, and/or physical function following survival of critical illness], although the incidence following COVID-19 is unknown.
International Literature
What does the international literature say?
Carfi, A et al (2020) Persistent Symptoms in Patients After Acute COVID-19[6]
The authors assessed persistent symptoms in patients who were discharged from hospital in Rome, Italy after recovery from COVID-19. 143 patients were included in the study. The mean age was 56.5 (SD, 14.6) years (range, 19-84 years), and 53 (37%) were women. During hospitalization, 72.7% of participants had evidence of interstitial pneumonia. The mean length of hospital stay was 13.5 (SD, 9.7) days; 21 patients (15%) received noninvasive ventilation and 7 patients (5%) received invasive ventilation.
Patients were assessed a mean of 60.3 (SD, 13.6) days after onset of the first COVID-19 symptom; at the time of the evaluation, only 18 (12.6%) were completely free of any COVID-19–related symptom, while 32% had 1 or 2 symptoms and 55% had 3 or more. None of the patients had fever or any signs or symptoms of acute illness. Worsened quality of life was observed among 44.1% of patients. In patients who had recovered from COVID-19, 87.4% reported persistence of at least 1 symptom, most often fatigue and dyspnea.
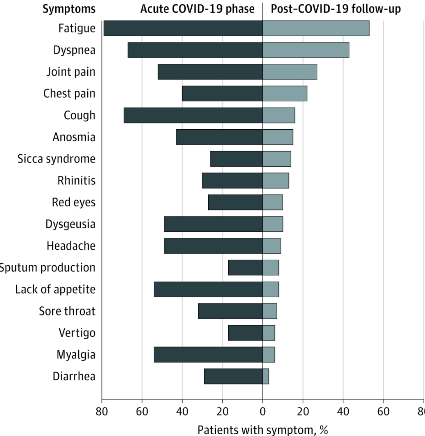
Prolonged symptom duration and disability are common in adults hospitalized with severe COVID-19. Characterizing return to baseline health among outpatients with milder COVID-19 illness is important for understanding the full spectrum of COVID-19-associated illness. During April 15-June 25, 2020, telephone interviews were conducted with a random sample of adults aged ≥18 years who had a first positive RT-PCR test for SARS-CoV-2, the virus that causes COVID-19, at an outpatient visit at one of 14 US academic health care systems in 13 states. Interviews were conducted 14-21 days after the test date. Respondents were asked about demographic characteristics, baseline chronic medical conditions, symptoms present at the time of testing, whether those symptoms had resolved by the interview date, and whether they had returned to their usual state of health at the time of interview.
Among 292 respondents, 94% (274) reported experiencing one or more symptoms at the time of testing; 35% of these symptomatic respondents reported not having returned to their usual state of health by the date of the interview (median 16 days from testing date), including 26% among those aged 18-34 years, 32% among those aged 35-49 years, and 47% among those aged ≥50 years. Among respondents reporting cough, fatigue or shortness of breath at the time of testing, 43%, 35%, and 29%, respectively, continued to experience these symptoms at the time of the interview. These findings indicate that COVID-19 can result in prolonged illness even among persons with milder outpatient illness, including young adults.
Greenhalgh, T et al (2020) Management of post-acute COVID-19 in primary care[8]
Management of COVID-19 after the first 3 weeks is currently based on limited evidence. Approximately 10% of people experience prolonged illness after COVID-19. Many such patients recover spontaneously (if slowly) with holistic support, rest, symptomatic treatment, and gradual increase in activity. Home pulse oximetry can be helpful in monitoring breathlessness. Indications for specialist assessment include clinical concern along with respiratory, cardiac or neurological symptoms that are new, persistent, or progressive.

Rubin, R (2020) As Their Numbers Grow, COVID-19 ‘Long Haulers’ Stump Experts[9]
In a recent JAMA research letter5, 125 of 143 Italian patients ranging in age from 19 to 84 years still experienced physician-confirmed COVID-19–related symptoms an average of 2 months after their first symptom emerged. All had been hospitalized, with their stays averaging 2 weeks; 80% had not received any form of ventilation. Physicians at a Paris hospital recently reported that they saw an average of 30 post-acute COVID patients every week between mid-May and late July. The patients’ average age was 40 years, and women outnumbered men 4 to 1. As with SARS, many post-acute COVID-19 patients are health workers who had massive exposure to the virus early in the pandemic.
Overall, approximately 10% of people who have contracted COVID-19 experience prolonged symptoms, Greengalgh at al7 estimated in a recently published article on post-acute COVID-19 management.
Adults with severe illness who spend weeks in intensive care can experience long-lasting symptoms, but that is not unique to patients with COVID-19; what is unusual about long COVID is that many initially had mild to moderate symptoms that did not require hospitalization.
Why some previously healthy, often young adults have not fully recovered from the disease has stymied physicians. In a recent study of 292 individuals with the disease who did not require hospitalization, Tenforde et al6 found that more than a third hadn’t returned to their usual state of health 2 to 3 weeks after testing positive. The older the patients, the more likely they were to state that their pre–COVID-19 health hadn’t come back; even a quarter of the youngest age group, those aged 18-34 years, stated that they had not yet regained their health. SARS-CoV-2 infection may trigger long-lasting changes in the immune system, and in some organs, especially the lungs, those changes persist far past the point at which patients have stopped shedding the virus.
Just as acute COVID-19 has been found to affect every part of the body, so, apparently, do its persistent symptoms. In the study of Italian patients5, the most common symptoms reported at follow-up were fatigue, shortness of breath, joint pain and chest pain. None of the patients had a fever or other signs or symptoms of acute illness, but about 44% had a worsened quality of life.
91% of 640 respondents to the Body Politic COVID-19 Support Group online survey in spring 2020 stated that they hadn’t fully recovered and were, on average, on day 40 of symptoms. Most reported ongoing fatigue, chills and sweats, body aches, headaches, brain fog, and gastrointestinal issues. Anecdotally, some people have reported feeling better for days or weeks before relapsing with old or new symptoms.
During a July webinar, Dr Anthony Fauci noted that some long COVID symptoms such as brain fog and fatigue are highly suggestive of chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME). About 3 out of 4 people diagnosed with CFS/ME report that it began with what appeared to be an infection, often infectious mononucleosis caused by Epstein-Barr virus (EBV). Although EBV is a herpesvirus, not a coronavirus, it has been speculated that SARS-CoV-2 infection might reactivate latent EBV, triggering the fatigue. Several scientists point to autonomic nervous system dysregulation as the possible explanation for tachycardia, extreme fatigue, and other persistent symptoms. The system controls involuntary physiologic processes such as heart rate, blood pressure, respiration and digestion.
Many post-acute COVID patients never had laboratory confirmation of COVID-19, which may add to some health care professionals’ scepticism that their persistent symptoms have a physiological basis. Only about ¼ of the Body Politic survey’s respondents had tested positive for COVID-19, while nearly half were never tested. Many respondents state that they aren’t always taken seriously, and that there is potential gender bias in the interpretation of reported persistent symptoms: women with persistent symptoms are more likely than men to be viewed as dramatic and anxious.
Pero, A et al (2020) COVID-19: A Perspective from Clinical Neurology and Neuroscience[10]
While the long-term effects of COVID-19 are currently unknown, a meta-analysis of ICU-related delirium patients reported persistent cognitive impairment up to 18 months after hospital discharge. Some speculate that peripheral cytokines during antiviral responses may cause neuropsychiatric symptoms by eliciting neuroinflammatory responses. Studies suggest that cognitive deficits related to cytokine storm syndrome may last for a long period of time after initial events. For example, after the 1918 flu pandemic, Parkinson’s disease (PD) was recorded as a long-term effect of encephalitis lethargica related to the infection. HCoV-229E and HCoV-OC43 were detected in many patients suffering from various neurological diseases, including PD and multiple sclerosis. A strong connection exists between neuroinflammation and Alzheimer’s disease (AD); thus, it is possible that COVID-19-associated neuroinflammation may lead to neurodegenerative processes such as AD. Psychiatric illness and fatigue may also be a long-term effect of COVID-19, as it was for the SARS epidemic. 63% of SARS survivors in a Hong Kong hospital were called in for a survey 41 months after their recovery, which revealed that over 40% had active psychiatric illness, 40% experienced chronic fatigue, and 27% were diagnosed with chronic fatigue syndrome.
[-] (2020) Long COVID: let patients help define long-lasting COVID symptoms[11]
Breathlessness and fatigue are among the continuing and debilitating symptoms being reported by people with COVID-19 often months after the onset of the disease and after they have been declared recovered.
Researchers and clinicians have yet to agree on a name for these ongoing symptoms ¾ post-COVID syndrome, chronic COVID-19 and long COVID are used alternately; and are also calling for the definition of recovery from COVID-19 to be based on criteria that extend beyond just testing negative. Symptoms such as chest heaviness, breathlessness, muscle pains, palpitations and fatigue should also be considered.
In deciding how to act on long COVID, researchers and policymakers should take heed of what happened in the case of myalgic encephalomyelitis, also called chronic fatigue syndrome (CFS/ME). The condition shares some of the symptoms of long COVID, and people with CFS/ME struggled for many years to be recognized as having a serious and debilitating medical condition that needed specialized treatment and research. The difficulties faced by people with CFS/ME and their representatives resulted, in part, from the fact that the patient voice was marginalized, which contributed to delays in the condition being recognized. It seems that the WHO is listening. In August, the Director General told a meeting of COVID patient groups: “We have received your SOS. We have heard loud and clear that long COVID needs recognition, guidelines, research and ongoing patient narratives to shape the WHO response from here on.”
Lamprecht, B (2020) Gibst es ein Post-COVID-Syndrom [Is there a post-COVID syndrome?][12]
For critically ill COVID-19 patients, surviving the acute phase of the disease might only mean having overcome the first stage of a long and challenging path. Physical, cognitive and psychological consequences seem to be probable; however, do residual symptoms in patients who have returned to microbiological normalization constitute post-COVID syndrome and which symptoms are possible? Critically ill patients often sustain functional limitations over a long period after discharge from hospital. In most cases of COVID-19 it is too early to speculate a diagnosis of post-COVID syndrome. Symptoms would have to have lasted over a period of at least 6 months; therefore, only post-infection fatigue can be considered at this point in time. Furthermore, even if patients recover physically, they could be at particular risk of suffering from longer-term mental health problems or a perceived reduction in quality of life. Based on previous experience with acute respiratory distress syndrome (ARDS), there is sufficient evidence for the potential existence of post-COVID syndrome.
Mardani, M (2020) Post COVID syndrome[13]
COVID-19 may take weeks to overcome; however, in some patients, symptoms persist even after the original infection. Longer-term complications can include heart failure, neurological disease and lung disease. Symptoms can include fatigue and brain fog, which may be related to cytokines that cross the blood-brain-barrier and affect the brain. These symptoms may reflect post-viral syndrome associated with COVID-19. Difficulty in reading, insomnia, general myalgia, dry skin, and increased anxiety are other common symptoms.
After the [SARS] outbreak, some people did not return to work for almost 20 months having developed myalgic encephalomyelitis/chronic fatigue syndrome (CFS/ME). A subgroup of remitted patients is likely to experience long-term adverse effects similar to CFS/ME such as persistent fatigue, diffuse myalgia, depression, and non-restorative sleep.
According to post-mortem findings in COVID-19 cases, the olfactory pathway is the route by which the virus can cross the blood-brain-barrier and enter the hypothalamus. The perivascular space along the olfactory nerves that can pass the cribriform plate into the nasal mucosa is an important pathway involved in brain lymphatic drainage. The anosmia observed in some COVID-19 patients may be related to the effects of coronavirus affecting this pathway. Moreover, the virus disturbs lymphatic drainage from the microglia in the brain. Production of pro-inflammatory agents such as interferon-gamma and interleukin-7 is caused by disturbance of lymphatic drainage. The production of cytokines in the central nervous system can cause post-viral symptoms because pro-inflammatory cytokines cross the blood-brain-barrier in circumventricular organs such as the hypothalamus resulting in autonomic impairment that is presented as a high fever. Long-term consequences are dysregulated sleep-wake cycle, cognitive impairment, and profound unremitting anergia [CFS/ME representations]. Following the SARS outbreak, the number of COVID-19 patients may increase and cause a severe post-viral syndrome that is called post-COVID-19 syndrome in which long-term chronic fatigue is associated with post-exertional neuroimmune exhaustion.
Common symptoms such as cough, fever, dyspnea, musculoskeletal symptoms and anosmia have been seen in a large proportion of patients in a study in Italy. Among patients discharged from hospital, 12.6% had no symptoms of COVID-19, 32% had one or two symptoms, and 55% had three or more. Also, 53% had fatigue, 43% dyspnea, 27% joint pain, and 21% chest pain. According to this study, 87.4% had persistence of at least one symptom, especially fatigue and dyspnea.
According to the Centers for Disease Control and Prevention (CDC), ME/CF affects 2.5 million Americans. A significant minority are homebound, even bedbound. As with post-COVID syndrome, the majority of people announce that their disease started with an acute episode of infectious disease, mostly mononucleosis or flu. Several neurologic, metabolic, immunologic and other impairments have been reported but the cause of CFS/ME has not yet been identified and there is no effective treatment. The cardinal symptom is not only fatigue; a long-term relapse of exhaustion and cognitive impairment following activity is reported, which is generally called post-exertional malaise.
Reassurance, self-care and symptomatic control are considered in patients at risk of post-viral syndrome. Assessing the prevalence of fatigue-induced symptoms and alleviating the symptoms and improving the quality of life for those affected after COVID-19 should be considered as a priority.
Nath, A (2020) Long-Haul COVID[14]
Modern medicine has faced its biggest challenge from the smallest of organisms. It is becoming increasingly apparent that many patients who recovered from the acute phase of the SARS-CoV-2 infection have persistent symptoms. This includes clouding of mentation, sleep disturbances, exercise intolerance and autonomic symptoms. Some also complain of persistent low grade fever and lymphadenopathy. Although there are no peer-reviewed papers at the moment on these patients, many news articles have been written about the phenomenon, and there are social media groups with several thousand patients describing these symptoms. The illness is referred to as ‘long-haul COVID’ or ‘long-tail COVID.’ Many of these patients are health workers who had massive exposure to the virus early in the pandemic, and describe having symptoms for 100+ days.
| Table 1. Autonomic symptoms in long-haul COVID-19 |
| Tachycardia upon mild exercise or standing |
| Night sweats |
| Temperature dysregulation |
| Gastroparesis |
| Constipation or diarrhoea |
| Peripheral vasoconstriction |
Halpin, SJ et al (2020) Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation[15]
There is currently very limited information on the nature and prevalence of post-COVID-19 symptoms after hospital discharge. A purposive sample of 100 survivors discharged from a large University hospital were assessed 4 to 8 weeks after discharge by a multidisciplinary team of rehabilitation professionals using a specialist telephone screening tool designed to capture symptoms and impact on daily life. Participants were between 29 and 71 days (mean 48 days) post-discharge from hospital. Thirty-two participants required treatment in an intensive care unit (ICU group) and 68 were managed in hospital wards without needing ICU care (ward group). New illness-related fatigue was the most common reported symptom by 72% of participants in the ICU group and 60.3% in the ward group. The next most common symptoms were breathlessness (65.6% in ICU group and 42.6% in ward group) and psychological distress (46.9% in ICU group and 23.5% in ward group). There was a clinically significant drop in EQ5D [health-related quality of life] in 68.8% in the ICU group and in 45.6% in the ward group.
This is the first study from the United Kingdom reporting on post-discharge symptoms. We recommend planning rehabilitation services to manage these symptoms appropriately and maximize the functional return of COVID-19 survivors.
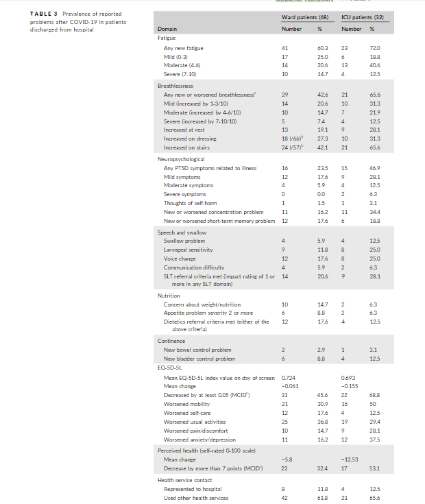
Objective: COVID-19 is a multisystem illness that has considerable long-term physical, psychological, cognitive, social and vocational sequelae in survivors. Given the scale of this burden and lockdown measures in most countries, there is a need for an integrated rehabilitation pathway using a tele-medicine approach to screen and manage these sequelae in a systematic and efficient way.
Methods: A multidisciplinary team of professionals in the UK developed a comprehensive pragmatic telephone screening tool, the COVID-19 Yorkshire Rehabilitation Screen (C19-YRS), and an integrated rehabilitation pathway, which spans the acute hospital trust, community trust and primary care service within the National Health Service (NHS) service model.
Results: The C19-YRS telephone screening tool, developed previously, was used to screen symptoms and grade their severity. Referral criteria thresholds were applied to the output of C19-YRS to inform the decision-making process in the rehabilitation pathway. A dedicated multidisciplinary COVID-19 rehabilitation team is the core troubleshooting forum for managing complex cases with needs spanning multiple domains of the health condition.
Conclusion: The authors recommend that health services dealing with the COVID-19 pandemic adopt a comprehensive telephone screening system and an integrated rehabilitation pathway to manage the large number of survivors in a timely and effective manner and to enable the provision of targeted interventions.
Marshall, M (2020) The lasting misery of coronavirus long-haulers[17]
Preliminary studies and existing research into other coronaviruses suggest that the virus can injure multiple organs and cause some surprising symptoms.
People with more severe infections might experience long-term damage not just in their lungs, but in their heart, immune system, brain and elsewhere. Evidence from previous coronavirus outbreaks, especially the severe acute respiratory syndrome (SARS) epidemic, suggests that these effects can last for years. And although in some cases the most severe infections also cause the worst long-term impacts, even mild cases can have life-changing effects – notably a lingering malaise similar to chronic fatigue syndrome.
Lung
A team at the University of Southern California analysed lung CT images of 919 patients from published studies and found that the lower lobes of the lungs are the most frequently damaged. The scans were riddled with opaque patches indicating inflammation that might make it difficult to breathe during sustained exercise. Visible damage normally reduced after two weeks. An Austrian study also found that lung damage lessened with time: 88% of participants had visible damage 6 weeks after being discharged from hospital, but by 12 weeks, the number had fallen to 56%.
A study posted on the preprint server medRxiv in August followed up on people who had been hospitalized and found that even a month after being discharged, more than 70% were reporting shortness of breath and 13.5% were still using oxygen at home.
Evidence from other coronaviruses suggests that lung damage may linger for some people. A study published in February recorded long-term lung harm caused by SARS-CoV-1. Between 2003 and 2018, Zhang et al minitored the health of 71 people who had been hospitalized with SARS. Even after 15 years, 4.6% still had visible lesions on their lungs, and 38% had reduced diffusion capacity.
Immune System
Some people who have recovered from COVID-19 could be left with a weakened immune system. The virus can also have the opposite effect, causing parts of the immune system to become overactive and trigger harmful inflammation throughout the body.
Heart
One organ particularly susceptible to inflammation is the heart. During the acute phase of COVID-19, about one-third of patients show cardiovascular symptoms. One such symptom is cardiomyopathy, in which the muscles of the heart become stretched, stiff or thickened, affecting the heart’s ability to pump blood. Some patients also have pulmonary thrombosis, in which a clot blocks a blood vessel in the lungs. The virus can also injure the wider circulatory system.
Brain
Many people who become severely ill experience neurological complications such as delirium, and there is evidence that cognitive difficulties, including confusion and memory loss, persist for some time after the acute symptoms have cleared; it is not clear whether complications are the result of the virus infecting the brain, or whether the symptoms are a secondary consequence — perhaps of inflammation.
Chronic Fatigue
One of the most insidious long-term effects of COVID-19 is its least understood: severe fatigue. Over the past nine months, an increasing number of people have reported crippling exhaustion and malaise after having the virus. Support groups on social media sites host thousands of members, who sometimes refer to [‘long-haul COVID’]. They struggle to get out of bed, or to work for more than a few minutes or hours at a time. One study of 143 people with COVID-19 discharged from a hospital in Rome found that 53% had reported fatigue and 43% had shortness of breath an average of 2 months after their symptoms started. A study of patients in China showed that 25% had abnormal lung function after 3 months, and that 16% were still fatigued.
Symptoms such as foggy thoughts, breathlessness and exhaustion resemble chronic fatigue syndrome, also known as myalgic encephalomyelitis (ME). The medical profession has struggled for decades to define CFS/ME leading to a breakdown of trust with some patients. There are no known biomarkers, so it can only be diagnosed based on symptoms. Because the cause is not fully understood, it is unclear how to develop a treatment. Dismissive attitudes from doctors persist, according to some patients. Fatigue does not seem to be limited to severe cases. It is common in people who had mild symptoms and who therefore might not have been tested for the virus.
Once again, there is evidence from SARS that coronavirus infection can cause long-term fatigue. In 2011, a stufy at the University of Toronto described 22 people with SARS, all of whom remained unable to work 13–36 months after infection. Compared with matched controls, they had persistent fatigue, muscle pain, depression and disrupted sleep. Another study, published in 2009, monitored people with SARS for 4 years and found that 40% had chronic fatigue. Many were unemployed and had experienced social stigmatization. A 2017 review of the literature on chronic fatigue syndrome found that many patients have persistent low-level inflammation, possibly triggered by infection. If COVID-19 is such a trigger, a wave of psychological effects “may be imminent”, write a group of researchers led by Declan Lyons, a psychiatrist at St Patrick’s Mental Health Services in Dublin. The ME Association in Britain says that it has received many reports of previously healthy people whose energy levels have not returned to normal after becoming infected with the virus, and expects to see new cases of chronic fatigue syndrome.
The current COVID-19 pandemic is not just a medical and social tragedy, but within the threat of the outbreak looms the potential for a significant and persistent negative mental health impact, based on previous experience with other pandemics such as Severe Acute Respiratory Syndrome (SARS) in 2003 and the earlier H1N1 outbreak of 1918. This article will highlight the links between depression and viral illnesses and explore important overlaps with myalgic encephalomyelitis/chronic fatigue syndrome (CFS/ME), potentially implicating inflammatory mechanisms in those exposed to a range of viral agents. While containment of psychological distress currently focuses on social anxiety and quarantine measures, a second wave of psychological morbidity due to viral illness may be imminent.
Fatigue is a common symptom in those presenting with symptomatic COVID-19 infection. However, it is unknown if COVID-19 results in persistent fatigue in those recovered from acute infection. We examined the prevalence of fatigue in individuals recovered from the acute phase of COVID-19 illness using the Chalder Fatigue Score (CFQ-11). We further examined potential predictors of fatigue following COVID-19 infection, evaluating indicators of COVID-19 severity, markers of peripheral immune activation and circulating pro-inflammatory cytokines. Of 128 participants (49.5 ± 15 years; 54% female), more than half reported persistent fatigue (52.3%; 45/128) at 10 weeks (median) after initial COVID-19 symptoms. There was no association between COVID-19 severity (need for inpatient admission, supplemental oxygen or critical care) and fatigue following COVID-19. Additionally, there was no association between routine laboratory markers of inflammation and cell turnover (leukocyte, neutrophil or lymphocyte counts, neutrophil-to-lymphocyte ratio, lactate dehydrogenase, C-reactive protein) or pro-inflammatory molecules (IL-6 or sCD25) and fatigue post COVID-19. Female gender and those with a pre-existing diagnosis of depression/anxiety were over-represented in those with fatigue. Our findings demonstrate a significant burden of post-viral fatigue in individuals with previous SARS-CoV-2 infection after the acute phase of COVID-19 illness. This study highlights the importance of assessing those recovering from COVID-19 for symptoms of severe fatigue, irrespective of severity of initial illness, and may identify a group worthy of further study and early intervention.
Donnelly, SC (2020) Post COVID Syndrome (PCS) and healthcare workers: who cares for the carers?[20]
Many of the author’s colleagues who have recovered from acute COVID-19 infection continue to suffer from a debilitating post-COVID syndrome (PCS), and are not able to return to work due to significant persistent emotional and physical disabilities.
Wise, J (2020) Long COVID: doctors call for research and surveillance to capture disease[21]
A group of doctors who have been affected by persistent symptoms of COVID-19 have written a manifesto based on their personal experiences. In a letter published in the BMJ, the group of 39 UK doctors called for research and surveillance to capture the full spectrum of disease, including in those not admitted to hospital and not tested, to build an accurate picture of COVID-19 phenotypes. They said that there was an emerging picture that prolonged symptoms were having substantial effects on a significant minority of people and that death was not the only outcome to measure. They called for a clear definition of recovery from COVID-19.
“Failure to understand the underlying biological mechanisms causing these persisting symptoms risks missing opportunities to identify risk factors, prevent chronicity, and find treatment approaches for people affected now and in the future.”
The action plan also called for the establishment of one-stop clinics involving multidisciplinary teams to help the rehabilitation of patients experiencing long COVID. Thorough investigations should be carried out to detect any organic pathology and to evaluate the patient’s cardiac and respiratory function before any active rehabilitation can start.
The doctors warned against reliance on a one-size-fits-all online rehabilitation service as it risks serious harm to patients if pathology goes undetected. They pointed out that the National Institute for Health and Care Excellence recently cautioned against using graded exercise therapy for patients recovering from COVID-19 and that it was important that such information was communicated quickly to clinicians.
Patients experiencing persisting symptoms of COVID-19 have a great deal to contribute to the search for solutions and should be involved in the commissioning of clinical services and the design of research studies.
Burgers, J (2020) Long COVID: the Dutch response[22]
Persisting symptoms after COVID-19 is also recognised as a serious problem in the Netherlands, and several actions have been taken to mitigate the problem. Rapid guidance has been developed to support clinicians in primary and secondary care in decision making about diagnosis and treatment. Because of a lack of evidence, most recommendations are based on expert opinion. The Dutch government has decided to reimburse physiotherapy, exercise therapy, ergotherapy and dietetics in primary care to a maximum of six months. The results of individual treatment will be monitored and evaluated systematically. Research has commenced on the follow-up of patients with COVID-19 funded by the Netherlands Organisation for Health Research and Development. One of the research aims is to identify predictors of chronicity.
Perrin, R et al (2020) Into the looking glass: Post-viral syndrome post COVID-19[23]
Post-mortem SARS research indicated that the virus had crossed the blood brain barrier into the hypothalamus via the olfactory pathway. The pathway of the virus seemed to follow that previously suggested in CFS/ME patients, involving disturbance of lymphatic drainage from the microglia in the brain. One of the main pathways of the lymphatic drainage of the brain is via the perivascular spaces along the olfactory nerves through the cribriform plate into the nasal mucosa. If the pathogenesis of coronavirus affects a similar pathway, it could explain the anosmia observed in a proportion of COVID-19 patients.
This disturbance leads to a build-up of pro-inflammatory agents, especially post-infectious cytokines such as interferon gamma, and interleukin 7, which have been hypothesized to affect the neurological control of the glymphatic system as observed in CFS/ME. The build-up of cytokines in the central nervous system may lead to post viral symptoms due to pro-inflammatory cytokines passing through the blood brain barrier in circumventricular organs such as the hypothalamus, leading to autonomic dysfunction manifesting acutely as a high fever and in the longer term to dysregulation of the sleep/wake cycle, cognitive dysfunction and profound unremitting anergia, all characteristic of CFS/ME. As happened after the SARS outbreak, a proportion of COVID-19 affected patients may go on to develop a severe post viral syndrome we term Post COVID-19 Syndrome: a long-term state of chronic fatigue characterised by post-exertional neuroimmune exhaustion.
Mahase, E (2020) COVID-19: What do we know about long COVID?[24]
Long COVID is a term being used to describe illness in people who have either recovered from COVID-19 but still report lasting effects of the infection or have had the usual symptoms for far longer than would be expected. Many people, including doctors who have been infected, have shared their anecdotal experiences on social media, in the traditional media, and through patients’ groups.
The Royal College of General Practitioners states: “There will be a significant influx of patients with lingering long COVID illness, both physical and emotional, and GPs must have the necessary resources and support to care for patients and help them come to terms with and readjust to the aftermath.”
Aside from anecdotal evidence, there is as yet little research on this issue. However, it is being actively discussed within the research community. Writing in JAMA, a team of researchers from Italy reported that nearly nine in 10 patients (87%) discharged from a Rome hospital after recovering from COVID-19 were still experiencing at least one symptom 60 days after onset. They found that 13% of the 143 people were completely free of any symptoms, while 32% had one or two symptoms, and 55% had three or more. Although none of the patients had fever or any signs or symptoms of acute illness, many still reported fatigue (53%), dyspnoea (43%), joint pain (27%), and chest pain (22%). Two fifths of patients reported a worsened quality of life.
The team behind the UK COVID-19 Symptom Study app, which collects symptom information from nearly four million users, says their data show that one in 10 people with COVID-19 are sick for greater than 3 weeks: “Most health sources suggest that people will recover within two weeks or so. But it’s becoming increasingly clear that this isn’t the case for everyone infected with coronavirus.”
After recovery from COVID-19, patients remain at risk for lung disease, heart disease, frailty and mental health disorders. There may also be long-term sequelae of adverse events that develop in the course of COVID-19 and its treatment. These complications are likely to place additional medical, psychological and economic burdens on all patients, with lower-income individuals, the uninsured or underinsured, and individuals experiencing homelessness being most vulnerable. There needs to be a comprehensive plan for preventing and managing post-COVID-19 complications to mitigate their clinical, economic and public health consequences and to support patients experiencing delayed morbidity and disability as a result.
The author aims to discuss: 1. findings related to neurologic complications; 2. how they connect to and are bi-directionally impacted by bioimmunology; 3. how the combination of biological mechanisms impact and are impacted by psychosocial stressors; and 4. the importance of considering potential neurodegenerative consequences of COVID-19. Longitudinal studies on neuropathology and cognition are critical to avoiding premature conclusions related to long-term neurologic effects.
Post-intensive care syndrome (PICS) describes new or worsening physical, cognitive or mental impairments in a patient following critical illness or intensive care. The COVID-19 pandemic will likely result in many more patients with PICS and its associated health and economic challenges. Screening and assessment tools completed during hospitalization, at discharge and post discharge should be utilized to facilitate services and strategies to improve PICS outcomes for patient and their families.
Hopkins, C et al (2020) Managing long COVID: don’t overlook olfactory dysfunction[28]
Loss of smell and taste is one of the most prevalent symptoms of COVID-19 and a predictor of COVID status. A substantial proportion of patients experience at least partial recovery of their loss of smell, but 10% report persistent loss eight weeks after onset. Parosmia ¾ distortion of the sense of smell that can severely impair appetite owing to familiar foods triggering a foul smell ¾ also seems to be prevalent in patients with COVID-19. [Parosmia] had already been reported by more than 50% of patients with post-viral olfactory loss before the COVID-19 pandemic. A similar disruption to normal life is likely to be found among patients with COVID-19.
A descriptive clinical follow-up (days 7, 30 [D30] and 60 [D60]) of 150 patients with non-critical COVID-19 confirmed by RT-PCR at Tours University Hospital from March 17 to June 3, 2020, including demographic, clinical and laboratory data collected from electronic medical records and by phone call. Persisting symptoms were defined by the presence at D30 or D60 of at least one of the following: weight loss ≥ 5%, severe dyspnea or asthenia, chest pain, palpitations, anosmia/ageusia, headache, cutaneous signs, arthralgia, myalgia, digestive disorders, fever or sick leave.
At D30, 68% (n=103/150) of patients presented at least one symptom and 66% (n=86/130) at D60, mainly anosmia/ageusia: 59% (n=89/150) at symptom onset, 28% (n=40/150) at D30 and 23% (n=29/130) at D60. Dyspnea concerned 36.7% (n=55/150) of patients at D30 and 30% (n=39/130) at D60. Half of the patients (n=74/150) at D30 and 40% (n=52/130) at D60 reported asthenia. Persistent symptoms at D60 were significantly associated with age, hospital admission and abnormal auscultation at symptom onset. At D30, severe COVID-19 and/or dyspnea at symptom onset were additional factors associated with persistent symptoms.
Up to 2 months after symptom onset, two thirds of adults with non-critical COVID-19 had complaints, mainly anosmia/ageusia, dyspnea or asthenia. A prolonged medical follow-up of patients with COVID-19 seems essential regardless of the initial clinical presentation.
Background: COVID-19 is defined by an association of multiple symptoms, including frequently reported olfactory and gustatory disorders. Objective: To analyze the prevalence of these neurosensory impairments in patients with COVID-19, and to assess short-term recovery. Methods: A multi-center case series study during the COVID-19 epidemic. All patients presenting a RT-PCR-confirmed SARS-CoV-2 infection were included, whether hospitalized or treated at home. To analyze the prevalence and features of olfactory and gustatory dysfunctions, a phone interview was conducted 5 days after the positive PCR result. A questionnaire was submitted again 10 days later to patients having reported olfactory and gustatory disorders in order to assess their recovery. Results: 115 patients were included in our study. 81 patients (70%) reported olfactory and gustatory disorders without nasal obstruction or rhinorrhea. These impairments were more frequently reported in the female population, young people, and house-bound patients with mild symptomatic forms. Short-term recovery assessed at D15 was complete for 64% of the patients, and incomplete in 33%. Median recovery time was 15 days (4-27 days) after olfactory or gustatory symptom onset. Conclusion: Olfactory and gustatory dysfunctions related to COVID-19 are frequently reported and prevalent in mild symptomatic forms of the disease. Recovery in most cases seems rapid and complete.
Dasgupta, A et al (2020) Long term complications and rehabilitation of COVID-19 patients[31]
Our previous experience with members of the same coronavirus family [SARS and MERS] which have caused two major epidemics in the past has given us insight that the harmful effects of such outbreaks are not limited to acute complications. Long-term cardiopulmonary, glucometabolic and neuropsychiatric complications have been documented following these infections. In the given circumstance it is therefore imperative to keep in mind the possible complications that may occur after the acute phase of the disease subsides and to prepare health systems for such challenges.
National Institute for Health Research (2020) Living with COVID-19[32]
This rapid and dynamic review draws on the lived experience of patients and expert consensus as well as published evidence to better understand the impact of ongoing effects of COVID-19, how health and social care services should respond, and what future research questions might be. The steering group concluded:
- There is a widespread perception that people either die, get admitted to hospital or recover after two weeks. It is increasingly clear that for some people there is a distinct pathway of ongoing effects. There is an urgent need to better understand the symptom journey and underlying clinical risks. People, their families and healthcare professionals need realistic expectations about what to expect.
- A major obstacle is the absence of consensus on diagnostic criteria for ongoing COVID-19. A working diagnosis that is recognised by healthcare services, employers and government agencies would facilitate access to much needed support and provide the basis for planning appropriate services. Although it is too early to give a precise definition, guidance on reaching a working diagnosis and a code for clinical datasets is needed.
- The fluctuating and multisystem symptoms need to be acknowledged. A common theme is that symptoms arise in one physiological system then abate only for symptoms to arise in a different system.
- There are significant psychological and social impacts that will have long-term consequences for individuals and for society if not well managed.
- The multisystem nature of ongoing COVID-19 means that it needs to be considered holistically, both in service provision and in research. The varying degrees of dependency mean support in the community should be considered alongside hospital one-stop clinics. Social support needs to be understood together with the financial pressures on previously economically active people.
- COVID-19 has a disproportionate effect on certain parts of the population, including care home residents. Black and Asian communities have seen high death rates and there are concerns about other minority groups and the socially disadvantaged.
Fjaeldstad, AW (2020) Prolonged complaints of chemosensory loss after COVID-19[33]
Note: Chemosensory loss: loss of smell [olfactory loss] and/or loss of taste [gustatory loss].
Objective: To investigate the temporal dynamics of improvement and recovery from sudden olfactory and gustatory loss in patients with confirmed and suspected COVID-19. Results: Among 109 study participants, 95 had a combined olfactory and gustatory loss, 5 participants had isolated olfactory loss and 9 participants has isolated taste loss. The mean age of participants was 39.4 years and 25% of participants were under the age of 30 years. Young age was not associated with a higher recovery rate. After a mean of > 30 days since chemosensory loss, participants reported relatively low recovery and improvement rates. For participants with olfactory loss, only 44% had fully recovered; 28% had not yet experienced any improvement of symptoms. After gustatory loss, 50% had fully recovered; 20% had not yet experienced any improvement. Olfactory and gustatory deficits were predominantly quantitative and mainly included complete loss of both olfactory and gustatory function.
Conclusion: Chemosensory loss was frequent in young individuals and persisted > 1 month after symptom onset, often without any improvement.
The authors included 120 patients after a mean (±SD) of 110.9 (±11.1) days following admission. The most frequently reported persistent symptoms were fatigue (55%), dyspnoea (42%), loss of memory (34%), concentration and sleep disorders (28% and 30.8%, respectively). Comparisons between ward and ICU patients led to no statistically significant differences regarding those symptoms. In both group, EQ-5D [mobility, self-care, pain, anxiety or depression, usual activity] was altered with a slight difference in pain in the ICU group.
The authors concluded that many patients requiring hospitalization for COVID-19 have persistent symptoms, and that findings should be confirmed in larger cohorts, including more severe patients.
Islam, MF et al (2020) ‘Post-viral fatigue and COVID-19: lessons from past epidemics[35]
The authors carried out a literature review on short- and long-term health consequences of prior epidemics and infections to assess potential health complications that may be associated with post-COVID-19 recovery. Past research on post-epidemic and post-infection recovery suggests that such complications include the development of severe fatigue. Certain factors such as the severity of infection in addition to the cytokine storm experienced by many COVID-19 patients may contribute to the development of later health problems. The authors suggest that the patterns observed in past epidemics and infections may re-occur in the current COVID-19 pandemic.
Kemp, HI et al (2020) Chronic pain after COVID-19: implications for rehabilitation[36]
The treatment needs of COVID-19 survivors are not yet fully appreciated. Although initially assumed to be a respiratory disease, it is now clear that it affects a variety of systems. Multi-organ failure may occur, with reports of cardiac, renal, haematological and neurological effects in the acute stages. These survivors will have significant multi-domain impairment requiring ongoing support. There has been a recent call to action among the rehabilitation community to ensure adequate resources to provide early phase, multidisciplinary interventions to promote physical and psychological recovery.
The COVID-19 pandemic is having a profound impact on the provision of medical care. As the curve progresses and patients are discharged the rehabilitation wave brings a high number of post-acute COVID-19 patients suffering from physical, mental and cognitive impairments impeding their return to normal life. The complexity and severity of disease in patients recovering from severe COVID-19 infection require an approach that is implemented as early in the recovery phase as possible, in a concerted and systematic way. To address the rehabilitation wave, the authors describe a spectrum of interventions that start in the ICU and continue through all appropriate levels of care. This approach requires organized rehabilitation teams including physiotherapists, occupational therapists, speech-language pathologists, rehabilitation psychologists/neuropsychologists and physiatrists collaborating with acute medical teams.
Background: An unknown proportion of people who had an apparently mild COVID-19 infection continue to suffer with persistent symptoms, including chest pain, shortness of breath, muscle and joint pains, headaches, cognitive impairment (brain fog), and fatigue. Post-acute COVID-19 or long-COVID seems to be a multisystem disease, sometimes occurring after a mild acute illness. Objective: To explore experiences of people with persisting symptoms following COVID-19 infection and to elicit their views on primary care support received. Design and Setting: Qualitative methodology, with semi-structured interviews to explore perspectives of people with persisting symptoms following suspected or confirmed COVID-19 infection. Participants were recruited via social media in July-August 2020. Method: Interviews were conducted by telephone or video call, digitally recorded, and transcribed with consent. Thematic analysis was conducted applying constant comparison techniques. People with experience of persisting symptoms contributed to study design and data analysis. Results: The main themes include: 1. the ‘hard and heavy work’ of enduring and managing symptoms and accessing care; 2. living with uncertainty, helplessness and fear, particularly over whether recovery is possible; 3. the importance of finding the ‘right’ GP [understanding, empathy, and support is needed]; and 4. recovery and rehabilitation. Conclusion: Patients require their GP to believe their symptoms and to demonstrate empathy and understanding. Ongoing support by primary care professionals during recovery and rehabilitation is crucial.
Tolba, M et al (2020) Assessment and Characterization of Post-COVID-19 manifestations[39]
287 survivors from COVID-19 were included in the study. Each received a questionnaire divided into: 1. demographic data; 2 COVID-19 status and other comorbidities; and 3. post-COVID-19 manifestations.
Only 10.8 % of all subjects had no manifestation after recovery from the disease while a large %age of subjects suffered from several symptoms. The most common symptom reported was fatigue (72.8 %). There was a relationship between the presence of other comorbidities and severity of the disease. Also, the severity of COVID-19 was related to the severity of post-COVID-19 manifestations.
Prevalence of anxiety or depression was investigated in 105 COVID-19 patients at 1 to 3 months from virological clearance by the HADS-A/D scale. 30% of patients displayed pathological HADS-A/D; 52.4% showed persistent symptoms. Pathological patients with HADS-A/D more commonly reported symptom persistence, even after adjustment for age, gender and disease severity. Psychological assessments should be encouraged in COVID-19 patients’ follow-up.
Tony, AA et al (2020) COVID-19-associated sleep disorders: A case report[41]
The authors reported one patient infected with acute respiratory syndrome coronavirus-2 (SARS-CoV-2) who presented with sleep disorders, insomnia and restless leg syndrome.
Vaes, AW et al (2020) Care Dependency in Non-Hospitalized Patients with COVID-19[42]
Background: A large sample of mild COVID-19 patients still experience multiple symptoms months after being infected. These persistent symptoms are associated with many clinically relevant outcomes, including poor health status and impaired functional status. To date, no information is available about care dependency. The authors aimed to explore the level of care dependency and the need for assistance with personal care in non-hospitalized COVID-19 patients. Methods: Members of social media groups for COVID-19 patients with persistent complaints in The Netherlands and Belgium were assessed for demographics, pre-existing comorbidities, health status, and symptoms. In addition, patients were asked about their dependence on others for personal care before and after the infection. The level of care dependency was assessed with the Care Dependency Scale (CDS) in members of the Belgian group (n = 210). Results: The data of 1837 non-hospitalized patients (86% women; median (IQR) age: 47 (38-54)) were analyzed. Only a small proportion of patients needed help with personal care before COVID-19, but the care need increased significantly after the infection (on average 79 ± 17 days after the onset of symptoms; 7.7% versus 52.4%, respectively; p < 0.05). The patients had a median (IQR) CDS score of 72 (67-75) points, and 31% of the patients were considered as care-dependent (CDS score ≤ 68 points).
Conclusions: COVID-19 has an important impact on care dependency in non-hospitalized patients. About 3 months after the onset of symptoms, a considerable proportion of non-hospitalized patients were to some degree dependent on others for personal care, indicating that the impact of COVID-19 on patients’ daily lives is significant, and more attention is needed to identify optimal treatment strategies to restore patients’ independence.
A multicentre prospective study on 138 COVID-19 patients. Olfactory and gustatory functions were prospectively evaluated for 60 days.
Within the first 4 days of disease, 84.8 % of patients had chemosensitive dysfunction that gradually improved over the observation period. The most significant increase in chemosensitive scores occurred in the first 10 days for taste and between 10 and 20 days for smell. At the end of the observation period (60 days after symptom onset), 7.2 % of patients still had severe dysfunctions. The risk of developing a long-lasting disorder becomes significant at 10 days for taste (odds ratio = 40.2, 95 % confidence interval = 2.204-733.2, p = 0.013) and 20 days for smell (odds ratio = 58.5, 95 % confidence interval = 3.278-1043.5, p = 0.005).
Chemosensitive disturbances persisted in 7.2 % of patients 60 days after clinical onset. Specific therapies should be initiated in patients with severe olfactory and gustatory disturbances 20 days after disease onset.
Objective: To investigate clinical outcomes, distribution of quarantine locations and infection status of the contacts of COVID-19 patients after discharge. Design: A prospective cohort study. Methods: Demographics and baseline characteristics of 131 COVID-19 patients discharged from 3 February 2020 to 21 February 2020 in Wuhan, China were collected and analyzed by reviewing medical records retrospectively. Post-hospitalization data relating to clinical outcomes, quarantine locations and close contact history were obtained by following up the patients every week for up to 4 weeks.
Results: On discharge, patients had cough (29.01%), fatigue (7.63%), expectoration (6.11%), chest tightness (6.11%), dyspnea (3.82%), chest pain (3.05%) and palpitations (1.53%). These symptoms constantly declined in the 4 weeks post-discharge. Transient fever recurred in 11 patients (8.4%). Among discharged patients, 78 (59.5%) underwent chest CT and 2 (1.53%) showed deterioration. A total of 94 (71.8%) patients received a SARS-CoV-2 retest, of which 8 (6.10%) reported positive. 7 (2.29%) patients were re-admitted because of fever or positive SARS-CoV-2 retest. After discharge, 121 (92.37%) and 4 (3.05%) patients were self-quarantined at home or in community facilities, respectively, after a close contact.
Conclusion: The majority of COVID-19 patients were in the course of recovery post-discharge. Readmission was required in rare cases due to suspected recurrence of COVID-19. Appropriate self-quarantine and regular re-examination are recommended, particularly for those with recurring symptoms.
Wilson, C (2020) Concern coronavirus may trigger post-viral fatigue syndromes[45]
An outbreak of post-viral fatigue syndrome in some people who have had COVID-19 is anticipated. Viral infections have previously been linked to problems with long-term fatigue symptoms: eg chronic fatigue syndrome/myalgic encephalomyelitis (CFS/ME). People who have CFS experience extreme fatigue and a range of other symptoms such as pain and sensitivity to light, but the condition is poorly understood. Although the current COVID-19 pandemic is caused by a different virus, it is a member of the same coronavirus family, so it might also give rise to post-viral fatigue syndrome.
Objective: To: 1. compare physical function and fitness outcomes in people infected with SARS-CoV with healthy controls; 2. quantify the recovery of physical function and fitness following SARS-CoV infection; and 3. determine the effects of exercise following SARS-CoV infection. Methods: Observational studies or randomized controlled trials were included if they involved people following SARS-CoV infection and either assessed the change or recovery in physical function/fitness or evaluated the effects exercise post-infection. Results: Evidence from 9 studies demonstrated that SARS-CoV patients had reduced levels of physical function and fitness post-infection compared with healthy controls. Furthermore, patients demonstrated incomplete recovery of physical function, with some experiencing residual impairments 1 to 2 years post-infection. Evidence from 1 randomized controlled trial found that a combined aerobic and resistance training intervention significantly improved physical function and fitness post-infection compared with a control group.
Conclusion: Physical function and fitness are impaired following SARS-CoV infection, and impairments may persist up to 2 years post-infection. Although one study demonstrated that exercise can improve physical function and fitness post-infection, further research is required to determine the effectiveness of exercise in people recovering from similar infections such as COVID-19.
Impact: Considering the similarities in pathology and clinical presentation of SARS-CoV and COVID-19, it is likely that COVID-19 patients will present with similar impairments to physical function. Accordingly, research is required to measure the extent of functional impairments in COVID-19 cohorts. In addition, research should evaluate whether rehabilitation interventions such as exercise can promote post-infection recovery.
Graded exercise therapy may not be appropriate for treating post-viral fatigue in patients recovering from COVID-19, the National Institute for Health and Care Excellence (NICE) has told doctors.
In a statement NICE said that it was aware of concerns related to the impact of graded exercise therapy for managing post-viral fatigue in patients recovering from COVID-19. NICE noted that its current advice on managing chronic fatigue may not be appropriate for COVID-19 patients and acknowledged that it could also be out of date for other groups.
Recently published COVID-19 research focuses more on describing the clinical presentations and the natural history of the pathology, rather than rehabilitation interventions or service delivery. Studies with high levels of evidence regarding the efficacy of interventions, long-term monitoring, or new organization models are needed.
Ahmed, H (2020) [Systematic Review] Long-term clinical outcomes in survivors of severe acute respiratory syndrome and Middle East respiratory syndrome coronavirus outbreaks after hospitalisation or ICU admission: A systematic review and meta-analysis[49]
Objective: To determine long-term clinical outcomes in survivors of severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS) coronavirus infections after hospitalization or intensive care unit admission. Study Selection: Original studies reporting clinical outcomes of adult SARS and MERS survivors 3 months after admission or 2 months after discharge were included in the systematic review. Data Synthesis: Of 1,169 identified studies, 28 were included in the analysis. Pooled analysis revealed that common complications up to 6 months after discharge were: impaired diffusing capacity for carbon monoxide (prevalence 27%, 95% confidence interval (CI) 15–45%); and reduced exercise capacity (mean 6-min walking distance 461 m, CI 450–473 m). The prevalence of post-traumatic stress disorder (39%, 95% CI 31–47%), depression (33%, 95% CI 20–50%) and anxiety (30%, 95% CI 10–61) beyond 6 months after discharge were considerable. Low scores on Short-Form 36 were identified beyond 6 months after discharge.
Conclusion: Lung function abnormalities, psychological impairment and reduced exercise capacity were common in SARS and MERS survivors. Clinicians should anticipate and investigate similar long-term outcomes in COVID-19 survivors.
Lee, SH and Shin, HS (2019) Depression as a Mediator of Chronic Fatigue and Post-Traumatic Stress Symptoms in Middle East Respiratory Syndrome Survivors[50]
The relationship among chronic fatigue, depressive symptoms and post-traumatic stress symptoms (PTSSs) in Middle East respiratory syndrome (MERS) survivors is poorly understood. Of 148 survivors who consented to be registered and underwent assessments at 12 months (T1) and 18 months (T2) after the MERS outbreak, 72 (48.65%) were evaluated for chronic fatigue, depressive symptoms and PTSSs based on the Impact of Event Scale (IES-R), the Patient Health Questionnaire-9 (PHQ-9), and the Fatigue Severity Scale (FSS). Data from 52 subjects who completed both assessments were analyzed using a regression-based serial multiple mediation model (PROCESS Model 6). Analyses indicated no direct effects of T1 FSS on T2 IES-R but significant positive indirect effects of T1 FSS on T2 IESR through T1 PHQ-9 and T2 PHQ-9 (B=2.1601, SE=1.3268, 95% confidence interval=0.4250-6.1307). In other words, both T1 PHQ-9 and T2 PHQ-9 fully mediated the relationship between T1 FSS and T2 IES.
Conclusion: Chronic fatigue 12 months after MERS had indirect effects on prolonged PTSSs 18 months after MERS via persisting depression in MERS survivors. This finding supports the need to promote interventional programs for emerging infectious disease survivors with chronic fatigue to reduce depression and prevent prolonged PTSSs.
Of 369 SARS survivors, 233 (63.1%) participated in the study (mean period of time after SARS, 41.3 months). Over 40% of the respondents had active psychiatric illnesses, 40.3% reported a chronic fatigue problem, and 27.1% met the modified 1994 Centers for Disease Control and Prevention criteria for chronic fatigue syndrome. Logistic regression analysis suggested that being a health care worker at the time of SARS infection (odds ratio [OR], 3.24; 95% confidence interval [CI], 1.12- 9.39; P = .03), being unemployed at follow-up (OR, 4.71; 95% CI, 1.50-14.78; P = .008), having a perception of social stigmatization (OR, 3.03; 95% CI, 1.20-7.60; P = .02), and having applied to the SARS survivors’ fund (OR, 2.92; 95% CI, 1.18-7.22; P = .02) were associated with an increased risk of psychiatric morbidities at follow-up. Application to the SARS survivors’ fund (OR, 2.64; 95% CI, 1.07-6.51; P = .04) was associated with increased risk of chronic fatigue problems.
Conclusions: Psychiatric morbidities and chronic fatigue persisted and continued to be clinically significant among SARS survivors at 4-year follow-up. Optimization of the treatment of mental health morbidities by a multidisciplinary approach with a view to long-term rehabilitation, especially targeting psychiatric and fatigue problems and functional and occupational rehabilitation, are indicated.
Other
The National Institute for Health and Care Excellence (NICE) and the Scottish Intercollegiate Guidelines Network (SIGN) have announced [on 5 October 2020] that they will work with the Royal College of General Practitioners to develop a guideline on persistent effects of COVID-19 (‘long COVID’) on patients.
The authors recommend that a 4-tier clinical service be developed for the management of long-COVID: Tier 1: resources and support for self-care; Tier 2: generalist care including a therapeutic relationship in general practice and a community-based interdisciplinary rehabilitation service led by health and social care professionals; Tier 3: specialist care including system-based investigation, management and rehabilitation; Tier 4: specialist management of specific complications.
Nabavi, N (2020) [Webinar] Long COVID: How to define it and how to manage it[54]
On 3 September the BMJ hosted an online webinar on the diagnosis, management and prognosis of long COVID. An expert panel discussed its symptoms, course and character, and suggested strategies for its management.
COVID Symptom Study (2020) [Website] How long does COVID-19 last?[55]
Data from the COVID Symptom Study suggests that while most people recover from COVID-19 within 2 weeks, one in ten people may still have symptoms after 3 weeks, and some may suffer for months.
Some people are experiencing fatigue, headaches, coughs, anosmia [loss of smell], sore throats, delirium and chest pain for more than 3 weeks after first reporting symptoms.
The survey targeted patients with prolonged symptoms [>2 weeks], although it does include some responses from patients with shorter symptom duration. The survey was primarily distributed to the Body Politic COVID-19 Support Group. The survey received 640 responses and was open from April 21 to May 2, 2020.
The top 10 symptoms reported over the course of the 8 weeks ranked highest to lowest are: mild shortness of breath, mild tightness of chest, moderate fatigue, mild fatigue, chills or sweats, mild body aches, dry cough, elevated temperature, mild headache, and brain fog/concentration challenges.
Patients indicate that their first week of experiencing symptoms had milder/fewer symptoms than weeks 2 and 3. Gastrointestinal symptoms and chills/sweats seem to occur more often in weeks 1-2 and respiratory symptoms appear relatively consistent through weeks 3-4.
Respondents had a major decline in physical activity since contracting the virus. Before symptom onset, 67% of respondents said that they were very/moderately physically active. At the time of the survey, 65% reported now being sedentary/mostly sedentary.
Neurological symptoms are underreported in the media. These include brain fog, concentration challenges, memory loss, seizures, dizziness and problems with balance, and various forms of insomnia.
Royal College of Occupational Therapists (2020) [Website] How to manage post-viral fatigue after COVID-19: Practical advice for people who have recovered at home[57]
Post-viral fatigue is when you have an extended period of feeling unwell and fatigued after a viral infection. Fatigue is a normal part of the body’s response to fighting a viral infection such as COVID-19. Fatigue is likely to continue for some time after the infection has cleared; it can make you sleep more, feel unsteady on your feet, make standing for long periods difficult, as well as affecting your ability to concentrate and your memory. This guide outlines practical advice to help combat fatigue.
Royal College of Occupational Therapists (2020) [Website] How to conserve your energy: Practical advice for people during and after having COVID-19[58]
More advice on ways to conserve energy after having COVID-19.
NHS England (2020) COVID-19: Supporting your recovery[59]
Although hospital admission is referred to throughout this resource, it can also be used for patients who remain in their own homes and will assist all patients and their families starting their rehabilitation journey following COVID-19.
Cox, D (2020) [News Article] Long COVID: the evidence of lingering heart damage: Cardiologists are finding that problems aren’t related to age or severity of infection[60]
Outlines stories of different patients who recovered from COVID-19 but still have lingering heart damage.
ScienceAlert.com (2020) [Website] Even People With Mild COVID-19 Symptoms Are Experiencing Long-Term Fatigue[61]
People who have been seriously unwell and treated in ICU can expect to take some months to recover fully, regardless of their ailment. However, with COVID-19, evidence is mounting that some people who have had relatively mild symptoms at home may also have a prolonged illness. Overwhelming fatigue, palpitations, muscle aches, pins and needles and many more symptoms are being reported as after-effects of the virus. Around 10 % of the 3.9 million people contributing to the COVID Symptom Study app have effects lasting more than 4 weeks.
The author gives his own experiences of long COVID and describes how a self-help pacing group has assisted him in managing persisting symptoms. The self-help pacing group is a forum to share experiences and provide mutual support.
Includes discussion about the real life experiences of people recovering from mild COVID and an online support group set up to help people with lingering symptoms.
Donohue, H (2020) [News Article] Fatigued: The long road to recovery from COVID-19[64]
A study carried out by Trinity College Dublin and St James’s Hospital found that 52% of COVID-19 survivors reported persistent fatigue 10 weeks after infection. The preliminary study ¾ which has not yet been peer-reviewed ¾ also indicated that fatigue persisted in patients regardless of the severity of initial symptoms.
NHS England (2020) [Website] Your COVID Recovery: Still feeling the effects?[65]
An NHS website providing information on what to expect during the recovery of a COVID-19 patient after hospitalisation.
Forthcoming Research and Clinical Trials: A Selection
University of Leicester (2020) Post-Hospitalisation COVID-19 Study (PHOSP-COVID)[66]
The Post-Hospitalisation COVID-19 Study (PHOSP-COVID) will look into the long-term health impacts of COVID-19. Patients recruited to the study will be assessed using advanced imaging, data collection and analysis of blood and lung samples to give a comprehensive picture of the impact COVID-19 has had on longer-term health outcomes in Britain.
Long-Term Outcomes in Patients with COVID-19 [Clinical Trial, Recruiting][67]
The ongoing COVID-19 pandemic has caused a global increase in hospitalization for pneumonia with multi- organ disease. Survival from sepsis is associated with increased risk of mortality for at least 2 years. Therefore, substantial sequelae including new physical disability, new cognitive impairment and increased vulnerability to further health deterioration are likely to be seen in survivors of COVID-19. The investigators will assess the daily living status of survivors of COVID-19 using a range of measures.
The investigators will study how long the virus can survive in different parts of the body, antibody development, and post-infectious complications. The investigators hope that the findings of the study will allow medical and public health providers to make recommendations to better care for patients in the convalescent phase of COVID-19 infection.
LIINC is an observational, prospective study of individuals previously infected with SARS-CoV-2 who have recovered from acute illness. The overall objective of the study is to investigate the clinical consequences of SARS-CoV-2 infection. These include the pre-existence and development of medical conditions, measures of immune activation and inflammation, changes in immunologic function, and variability in host responses. There will be a specific focus on demographic differences including age, gender and race.
[1] World Health Organisation (September 2020) What we know about long term effects of COVID-19. The latest on the COVID-19 global situation and long-term sequelae. https://www.who.int/docs/default-source/coronaviruse/risk-comms-updates/update-36-long-term-symptoms.pdf?sfvrsn=5d3789a6_2 [Accessed 19 October 2020]
[2] World Health Organization (2020) Support for Rehabilitation Self-Management after COVID-19- Related Illness
https://www.who.int/publications/m/item/support-for-rehabilitation-self-management-after-COVID-19-related-illness
[Accessed 19 October 2020]
[3] National Institute for Health and Care Excellence (2020) COVID-19 Rapid Guideline: Managing the Long-Term Effects of COVID-19 https://www.nice.org.uk/guidance/NG188 [Accessed 22 December 2020]
[4] BMJ Best Practice (2020) Coronavirus disease 2019 (COVID-19). https://bestpractice.bmj.com/topics/en-gb/3000201/complications [Accessed 28 October 2020]
[5] UpToDate (2020) Coronavirus Disease 2019 (COVID-19). https://www.uptodate.com/contents/coronavirus-disease-2019-covid-19-clinical-features [Accessed 28 October 2020]
[6] Carfì A, Bernabei R, Landi F; Gemelli Against COVID-19 Post-Acute Care Study Group. Persistent Symptoms in Patients After Acute COVID-19. JAMA. 2020 Aug 11;324(6):603-605. doi: 10.1001/jama.2020.12603. PMID: 32644129; PMCID: PMC7349096
[7] Tenforde MW, Kim SS, Lindsell CJ, Billig Rose E, Shapiro NI, Files DC, Gibbs KW, Erickson HL, Steingrub JS, Smithline HA, Gong MN, Aboodi MS, Exline MC, Henning DJ, Wilson JG, Khan A, Qadir N, Brown SM, Peltan ID, Rice TW, Hager DN, Ginde AA, Stubblefield WB, Patel MM, Self WH, Feldstein LR; IVY Network Investigators; CDC COVID-19 Response Team; IVY Network Investigators. Symptom Duration and Risk Factors for Delayed Return to Usual Health Among Outpatients with COVID-19 in a Multistate Health Care Systems Network – United States, March-June 2020. MMWR Morb Mortal Wkly Rep. 2020 Jul 31;69(30):993-998. doi: 10.15585/mmwr.mm6930e1. PMID: 32730238; PMCID: PMC7392393
[8] Greenhalgh T, Knight M, A’Court C, Buxton M, Husain L. Management of post-acute COVID-19 in primary care. BMJ. 2020 Aug 11;370:m3026. doi: 10.1136/bmj.m3026. PMID: 32784198
[9] Rubin R. As Their Numbers Grow, COVID-19 ‘Long Haulers’ Stump Experts. JAMA. 2020 Sep 23. doi: 10.1001/jama.2020.17709. Epub ahead of print. PMID: 32965460
[10][10] Pero A, Ng S, Cai D. COVID-19: A Perspective from Clinical Neurology and Neuroscience. Neuroscientist. 2020 Oct-Dec;26(5-6):387-391. doi: 10.1177/1073858420946749. Epub 2020 Jul 25. PMID: 32713251
[11] Long COVID: let patients help define long-lasting COVID symptoms. Nature. 2020 Oct;586(7828):170. doi: 10.1038/d41586-020-02796-2. PMID: 33029005
[12][12] Lamprecht B. Gibt es ein Post-COVID-Syndrom? [Is there a post-COVID syndrome?]. Pneumologe (Berl). 2020 Oct 8:1-4. German. doi: 10.1007/s10405-020-00347-0. Epub ahead of print. PMID: 33052198; PMCID: PMC7543035
[13] Mardani, M. Post COVID syndrome. Archives of Clinical Infectious Diseases 2020 15 (2). https://sites.kowsarpub.com/archcid/articles/108819.html. [Accessed 28 October 2020]
[14][14] Nath A. Long-Haul COVID. Neurology. 2020 Sep 29;95(13):559-560. doi: 10.1212/WNL.0000000000010640. Epub 2020 Aug 11. PMID: 32788251
[15] Halpin SJ, McIvor C, Whyatt G, Adams A, Harvey O, McLean L, Walshaw C, Kemp S, Corrado J, Singh R, Collins T, O’Connor RJ, Sivan M. Postdischarge symptoms and rehabilitation needs in survivors of COVID-19 infection: A cross-sectional evaluation. J Med Virol. 2020 Jul 30. doi: 10.1002/jmv.26368. Epub ahead of print. PMID: 32729939
[16] Sivan M, Halpin S, Hollingworth L, Snook N, Hickman K, Clifton IJ. Development of an integrated rehabilitation pathway for individuals recovering from COVID-19 in the community. J Rehabil Med. 2020 Aug 24;52(8):jrm00089. doi: 10.2340/16501977-2727. PMID: 32830284.
[17] Marshall M. The lasting misery of coronavirus long-haulers. Nature. 2020 Sep;585(7825):339-341. doi: 10.1038/d41586-020-02598-6. PMID: 32929257
[18] Lyons D, Frampton M, Naqvi S, Donohoe D, Adams G, Glynn K. Fallout from the COVID-19 pandemic – should we prepare for a tsunami of post viral depression? Ir J Psychol Med. 2020 May 15:1-6. doi: 10.1017/ipm.2020.40. Epub ahead of print. PMID: 32408926; PMCID: PMC7431842.
[19] Townsend, L et al (2020) Persistent fatigue following SARS-CoV-2 infection is common and independent of severity of initial infection. https://www.medrxiv.org/content/10.1101/2020.07.29.20164293v1. [Accessed 28 October 2020]
[20] Donnelly SC. Post COVID Syndrome (PCS) and healthcare workers: who cares for the carers? QJM. 2020 Sep 1;113(9):611. doi: 10.1093/qjmed/hcaa248. PMID: 32901263
[21] Wise J. Long COVID: doctors call for research and surveillance to capture disease. BMJ. 2020 Sep 15;370:m3586. doi: 10.1136/bmj.m3586. PMID: 32933925
[22] Burgers J. “Long COVID”: the Dutch response. BMJ. 2020 Aug 14;370:m3202. doi: 10.1136/bmj.m3202. PMID: 32816711
[23] Perrin R, Riste L, Hann M, Walther A, Mukherjee A, Heald A. Into the looking glass: Post-viral syndrome post COVID-19. Med Hypotheses. 2020 Jun 27;144:110055. doi: 10.1016/j.mehy.2020.110055. Epub ahead of print. PMID: 32758891; PMCID: PMC7320866
[24] Mahase E. COVID-19: What do we know about “long COVID”? BMJ. 2020 Jul 14;370:m2815. doi: 10.1136/bmj.m2815. PMID: 32665317
[25] Jiang DH, McCoy RG. Planning for the Post-COVID Syndrome: How Payers Can Mitigate Long-Term Complications of the Pandemic. J Gen Intern Med. 2020 Oct;35(10):3036-3039. doi: 10.1007/s11606-020-06042-3. Epub 2020 Jul 22. PMID: 32700223; PMCID: PMC7375754.
[26] Lennon JC. Neurologic and Immunologic Complications of COVID-19: Potential Long-Term Risk Factors for Alzheimer’s Disease. J Alzheimers Dis Rep. 2020 Jun 16;4(1):217-221. doi: 10.3233/ADR-200190. PMID: 32715280; PMCID: PMC7369139
[27] Biehl M, Sese D. Post-intensive care syndrome and COVID-19 – Implications post pandemic. Cleve Clin J Med. 2020 Aug 5. doi: 10.3949/ccjm.87a.ccc055. Epub ahead of print. PMID: 32759175.
[28] Hopkins C, Burges Watson DL, Kelly C, Leary V, Smith BC. Managing long COVID: don’t overlook olfactory dysfunction. BMJ. 2020 Sep 25;370:m3736. doi: 10.1136/bmj.m3736. Erratum in: BMJ. 2020 Sep 30;370:m3771. PMID: 32978178
[29] Carvalho-Schneider C, Laurent E, Lemaignen A, Beaufils E, Bourbao-Tournois C, Laribi S, Flament T, Ferreira-Maldent N, Bruyère F, Stefic K, Gaudy-Graffin C, Grammatico-Guillon L, Bernard L. Follow-up of adults with non-critical COVID-19 two months after symptoms’ onset. Clin Microbiol Infect. 2020 Oct 5:S1198-743X(20)30606-6. doi: 10.1016/j.cmi.2020.09.052. Epub ahead of print. PMID: 33031948; PMCID: PMC7534895
[30] Chary E, Carsuzaa F, Trijolet JP, Capitaine AL, Roncato-Saberan M, Fouet K, Cazenave-Roblot F, Catroux M, Allix-Beguec C, Dufour X. Prevalence and Recovery From Olfactory and Gustatory Dysfunctions in COVID-19 Infection: A Prospective Multicenter Study. Am J Rhinol Allergy. 2020 Sep;34(5):686-693. doi: 10.1177/1945892420930954. Epub 2020 Jun 12. PMID: 32527141; PMCID: PMC7418272.
[31] Dasgupta A, Kalhan A, Kalra S. Long term complications and rehabilitation of COVID-19 patients. J Pak Med Assoc. 2020 May;70(Suppl 3)(5):S131-S135. doi: 10.5455/JPMA.32. PMID: 32515393.
[32] NIHR. (2020) Living with COVID-19, National Institute for Health Research Centre for Engagement and Dissemination, available: https://evidence.nihr.ac.uk/themedreview/living-with-COVID-19. Accessed 19 10 2020.
[33] Fjaeldstad AW. Prolonged complaints of chemosensory loss after COVID-19. Dan Med J. 2020 Aug 1;67(8):A05200340. PMID: 32741438.
[34] Garrigues E, Janvier P, Kherabi Y, Le Bot A, Hamon A, Gouze H, Doucet L, Berkani S, Oliosi E, Mallart E, Corre F, Zarrouk V, Moyer JD, Galy A, Honsel V, Fantin B, Nguyen Y. Post-discharge persistent symptoms and health-related quality of life after hospitalization for COVID-19. J Infect. 2020 Aug 25:S0163-4453(20)30562-4. doi: 10.1016/j.jinf.2020.08.029. Epub ahead of print. PMID: 32853602; PMCID: PMC7445491.
[35] Islam, M.F., Cotler, J. and Jason, L.A. (2020) ‘Post-viral fatigue and COVID-19: lessons from past epidemics’, Fatigue: Biomedicine, Health and Behavior, 8(2), 61-69, available: http://dx.doi.org/10.1080/21641846.2020.1778227. Accessed 23 10 2020.
[36] Kemp HI, Corner E, Colvin LA. Chronic pain after COVID-19: implications for rehabilitation. Br J Anaesth. 2020 Oct;125(4):436-440. doi: 10.1016/j.bja.2020.05.021. Epub 2020 May 31. PMID: 32560913; PMCID: PMC7261464.
[37] Kim SY, Kumble S, Patel B, Pruski AD, Azola A, Tatini AL, Nadendla K, Richards L, Keszler MS, Kott M, Friedman M, Friedlander T, Silver K, Hoyer EH, Celnik P, Lavezza A, González-Fernández M. Managing the Rehabilitation Wave: Rehabilitation Services for COVID-19 Survivors. Arch Phys Med Rehabil. 2020 Sep 22:S0003-9993(20)30955-2. doi: 10.1016/j.apmr.2020.09.372. Epub ahead of print. PMID: 32971100; PMCID: PMC7506328.
[38] Kingstone T, Taylor AK, O’Donnell CA, Atherton H, Blane DN, Chew-Graham CA. Finding the ‘right’ GP: a qualitative study of the experiences of people with long-COVID. BJGP Open. 2020 Oct 13:bjgpopen20X101143. doi: 10.3399/bjgpopen20X101143. Epub ahead of print. PMID: 33051223.
[39] Tolba M, Abo Omirah M, Hussein A, Saeed H. Assessment and Characterization of Post-COVID-19 manifestations. Int J Clin Pract. 2020 Sep 29:e13746. doi: 10.1111/ijcp.13746. Epub ahead of print. PMID: 32991035; PMCID: PMC7536922.
[40] Tomasoni D, Bai F, Castoldi R, Barbanotti D, Falcinella C, Mulè G, Mondatore D, Tavelli A, Vegni E, Marchetti G, d’Arminio Monforte A. Anxiety and depression symptoms after virological clearance of COVID-19: A cross-sectional study in Milan, Italy. J Med Virol. 2020 Aug 25:10.1002/jmv.26459. doi: 10.1002/jmv.26459. Epub ahead of print. PMID: 32841387; PMCID: PMC7461061.
[41] Tony AA, Tony EA, Ali SB, Ezzeldin AM, Mahmoud AA. COVID-19-associated sleep disorders: A case report. Neurobiol Sleep Circadian Rhythms. 2020 Nov;9:100057. doi: 10.1016/j.nbscr.2020.100057. Epub 2020 Sep 12. PMID: 32954046; PMCID: PMC7486182.
[42] Vaes AW, Machado FVC, Meys R, Delbressine JM, Goertz YMJ, Van Herck M, Houben-Wilke S, Franssen FME, Vijlbrief H, Spies Y, Van ‘t Hul AJ, Burtin C, Janssen DJA, Spruit MA. Care Dependency in Non-Hospitalized Patients with COVID-19. J Clin Med. 2020 Sep 12;9(9):E2946. doi: 10.3390/jcm9092946. PMID: 32932582.
[43] Vaira LA, Hopkins C, Petrocelli M, Lechien JR, Chiesa-Estomba CM, Salzano G, Cucurullo M, Salzano FA, Saussez S, Boscolo-Rizzo P, Biglioli F, De Riu G. Smell and taste recovery in coronavirus disease 2019 patients: a 60-day objective and prospective study. J Laryngol Otol. 2020 Aug;134(8):703-709. doi: 10.1017/S0022215120001826. Epub 2020 Aug 12. PMID: 32782030; PMCID: PMC7471571.
[44] Wang X, Xu H, Jiang H, Wang L, Lu C, Wei X, Liu J, Xu S. Clinical features and outcomes of discharged coronavirus disease 2019 patients: a prospective cohort study. QJM. 2020 Sep 1;113(9):657-665. doi: 10.1093/qjmed/hcaa178. PMID: 32442308; PMCID: PMC7313792.
[45] Wilson C. Concern coronavirus may trigger post-viral fatigue
syndromes. New Sci. 2020 Apr 18;246(3278):10-11. doi: 10.1016/S0262-4079(20)30746-6. Epub 2020 Apr 17. PMID: 32372811; PMCID: PMC7194715.
[46] Rooney S, Webster A, Paul L. Systematic Review of Changes and Recovery in Physical Function and Fitness After Severe Acute Respiratory Syndrome-Related Coronavirus Infection: Implications for COVID-19 Rehabilitation. Phys Ther. 2020 Sep 28;100(10):1717-1729. doi: 10.1093/ptj/pzaa129. PMID: 32737507; PMCID: PMC7454932.
[47] Torjesen I. NICE advises against using graded exercise therapy for patients recovering from COVID-19. BMJ. 2020 Jul 21;370:m2912. doi: 10.1136/bmj.m2912. Erratum in: BMJ. 2020 Jul 22;370:m2933. PMID: 32694164.
[48] Andrenelli E, Negrini F, De Sire A, Patrini M, Lazzarini SG, Ceravolo MG; International Multiprofessional Steering Committee of Cochrane Rehabilitation REH-COVER action. Rehabilitation and COVID-19: a rapid living systematic review 2020 by Cochrane Rehabilitation Field. Update as of September 30th, 2020. Eur J Phys Rehabil Med. 2020 Oct 29. doi: 10.23736/S1973-9087.20.06672-1. Epub ahead of print. PMID: 33118719.
[49] Ahmed H, Patel K, Greenwood DC, Halpin S, Lewthwaite P, Salawu A, Eyre L, Breen A, O’Connor R, Jones A, Sivan M. Long-term clinical outcomes in survivors of severe acute respiratory syndrome and Middle East respiratory syndrome coronavirus outbreaks after hospitalisation or ICU admission: A systematic review and meta-analysis. J Rehabil Med. 2020 May 31;52(5):jrm00063. doi: 10.2340/16501977-2694. PMID: 32449782.
[50] Lee SH, Shin HS, Park HY, Kim JL, Lee JJ, Lee H, Won SD, Han W. Depression as a Mediator of Chronic Fatigue and Post-Traumatic Stress Symptoms in Middle East Respiratory Syndrome Survivors. Psychiatry Investig. 2019 Jan;16(1):59-64. doi: 10.30773/pi.2018.10.22.3. Epub 2019 Jan 7. PMID: 30605995; PMCID: PMC6354037.
[51] Lam MH, Wing YK, Yu MW, Leung CM, Ma RC, Kong AP, So WY, Fong SY, Lam SP. Mental morbidities and chronic fatigue in severe acute respiratory syndrome survivors: long-term follow-up. Arch Intern Med. 2009 Dec 14;169(22):2142-7. doi: 10.1001/archinternmed.2009.384. PMID: 20008700.
[52] National Institute for Health and Care Excellence (2020) NICE and SIGN announce latest rapid COVID-19 guideline will address Long COVID. https://www.nice.org.uk/news/article/nice-sign-announce-latest-rapid-COVID-19-guideline-will-address-long-COVID [Accessed 19 October 2020]
[53] Greenhalgh, T et al (2020) ‘Long COVID’: evidence, recommendations and priority research questions https://committees.parliament.uk/writtenevidence/12345/pdf/. [Accessed 19 October 2020]
[54] Nabavi N. Long COVID: How to define it and how to manage it. BMJ. 2020 Sep 7;370:m3489. doi: 10.1136/bmj.m3489. PMID: 32895219
[55] COVID Symptom Study (2020) [Website] How long does COVID-19 last? https://COVID.joinzoe.com/post/COVID-long-term [Accessed 19 October 2020]
[56] Assaf G, Davis H, McCorkell L, et al. An analysis of the prolonged COVID-19 symptoms survey by Patient-Led Research Team. Patient Led Research, 2020. https://patientresearchCOVID19.com/
[57] Royal College of Occupational Therapists (2020) How to manage post-viral fatigue after COVID-19: Practical advice for people who have recovered at home. https://www.rcot.co.uk/how-manage-post-viral-fatigue-after-COVID-19-0 [Accessed 19 October 2020]
[58] Royal College of Occupational Therapists (2020) How to conserve your energy: Practical advice for people during and after having COVID-19 https://www.rcot.co.uk/conserving-energy [Accessed 19 October 2020]
[59] NHS England (2020) COVID-19: Supporting your recovery
https://COVIDpatientsupport.lthtr.nhs.uk/#/ [Accessed 19 October 2020]
[60] Cox, D (2020) Long COVID: the evidence of lingering heart damage: Cardiologists are finding that problems aren’t related to age or severity of infection https://www.theguardian.com/world/2020/oct/04/long-COVID-the-evidence-of-lingering-heart-damage [Accessed 20 October 2020]
[61] ScienceAlert.com (2020) [Website] Even People With Mild COVID-19 Symptoms Are Experiencing Long-Term Fatigue
https://www.sciencealert.com/why-are-some-people-experiencing-long-term-fatigue-after-recovering-from-COVID-19 [Accessed 19 October 2020]
[62] Garner P et al (2020) [Blog] Long COVID and self-help pacing groups—getting by with a little help from our friends https://blogs.bmj.com/bmj/2020/09/29/long-COVID-and-self-help-pacing-groups-getting-by-with-a-little-help-from-our-friends/ [Accessed 22 October 2020]
[63] Brennan S (Sept 2020) [News Article] COVID and the brain: ‘This has taken almost everything that is me away from me’ https://www.irishtimes.com/life-and-style/health-family/COVID-and-the-brain-this-has-taken-almost-everything-that-is-me-away-from-me-1.4343793 [Accessed 22 October 2020]
[64] Donohue, H (2020) [News Article] Fatigued: The long road to recovery from COVID-19
https://www.rte.ie/news/coronavirus/2020/0928/1167831-COVID-long-term-effect/ [Accessed 19 October 2020]
[65] NHS England (2020) Your COVID Recovery: Still feeling the effects?
https://www.yourCOVIDrecovery.nhs.uk/ [Accessed 19 October 2020]
[66] University of Leicester (2020) Post-hospitalisation COVID-19 study (PHOSP-COVID). https://www.phosp.org/. [Accessed 28 October 2020]
[67] Long-Term Outcomes in Patients with COVID-19. https://clinicaltrials.gov/ct2/show/NCT04508712. [Accessed 28 October 2020]
[68] Determinants of SARS-COV2 (COVID-19) Persistence After Convalescence. https://clinicaltrials.gov/ct2/show/NCT04448145. [Accessed 28 October 2020]
[69] Long-term Impact of Infection With Novel Coronavirus (COVID-19) (LIINC) https://clinicaltrials.gov/ct2/show/NCT04362150 [Accessed 28 October 2020]
Produced by the members of the National Health Library and Knowledge Service Evidence Team†. Current as at 30 October 2020. This evidence summary collates the best available evidence at the time of writing and does not replace clinical judgement or guidance. Emerging literature or subsequent developments in respect of COVID-19 may require amendment to the information or sources listed in the document. Although all reasonable care has been taken in the compilation of content, the National Health Library and Knowledge Service Evidence Team makes no representations or warranties expressed or implied as to the accuracy or suitability of the information or sources listed in the document. This evidence summary is the property of the National Health Library and Knowledge Service and subsequent re-use or distribution in whole or in part should include acknowledgement of the service.
Brendan Leen, Area Library Manager, HSE South [Author, Editor]; Isabelle Delaunois, Librarian, University Hospital Limerick [Author]; Marie Carrigan, Librarian, St. Luke’s Radiation Oncology Network, Rathgar, Dublin [Author]; Siobhan McCarthy, Health Intelligence Unit, Strategic Planning and Transformation [Author]

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
