Are those with heart failure at greater risk of severe COVID-19 infection?
The following information resources have been selected by the National Health Library and Knowledge Service Evidence Virtual Team in response to a question from the National Immunisation Advisory Committee (NIAC). The resources are listed in our estimated order of relevance to practicing healthcare professionals confronted with this scenario in an Irish context. In respect of the evolving global situation and rapidly changing evidence base, it is advised to use hyperlinked sources in this document to ensure that the information you are disseminating to the public or applying in clinical practice is the most current, valid and accurate. For further information on the methodology used in the compilation of this document ⎯ including a complete list of sources consulted ⎯ please see our National Health Library and Knowledge Service Summary of Evidence Protocol.
DOWNLOAD FULL SUMMARY OF EVIDENCE (PDF)
Main Points
- Evidence from research studies and international guidelines indicates that pre-existing heart failure significantly increases the risk of severe COVID-19 and the risk of mortality.
- In patients with COVID-19, heart failure may be precipitated by acute illness, acute haemodynamic stress, or acute myocardial injury. Cardiovascular risk factors and cardiovascular disease are highly prevalent in hospitalized patients with COVID-19. Patients with a known history of heart failure may suffer an acute decompensation due to the development of COVID-19 disease.
- Severe clinical presentation and elevated biomarkers in COVID-19 patients with established risk factors can predict mortality from cardiovascular causes.
Summary of Evidence
Summary of Evidence
Evidence from research studies and international guidelines1-8 indicates that pre-existing heart failure significantly increases the risk of severe COVID-19 and the risk of mortality. Heart failure in patients with COVID-19 may be precipitated by acute illness with pre-existing known or undiagnosed heart disease, acute hemodynamic stress, or acute myocardial injury. Cardiovascular risk factors and cardiovascular disease are highly prevalent in hospitalized patients with COVID-19. Patients with a known history of heart failure may suffer an acute decompensation due to the development of COVID-19 disease8.
Apart from Hessami et al11 and Momtazmanesh et al13, the risk of bias was thought to be substantial in several of the included studies in the systematic reviews found, and results should be interpreted with caution.
Hessami et al11 shows that heart failure was not significantly associated with ICU admission in COVID-19. The authors also state that presence of acute cardiac injury, coronary artery disease, arrhythmia, hypertension, heart failure and cardiovascular disease were significantly associated with mortality in COVID-19 patients.
Based on their meta-analysis, Momtazmanesh et al13 conclude that there is a wide spectrum of cardiac involvement in COVID-19 patients. The authors recommend a triage risk stratification tool that could serve as a guide for the timely recognition of high-risk patients and mechanism-targeted therapy.
Chidambaram et al9 found that the risk of mortality was higher in patients with congestive heart failure. Clinically relevant cut-offs for leukocytosis, lymphopenia, elevated C-reactive protein LDH and D-dimer had higher odds of severe disease and greater risk of mortality. The systematic review presents some limitations: time points of evaluation of laboratory, radiological and disease severity assessment were not clearly defined in many of the studies, which precluded the calculation of risk ratio of severe disease; and the authors did not take into account variance in critical care interventions and strategies that potentially impact the course of the disease and/or survival.
Keeley et al12 summarized symptoms in patients with COVID-19, with a specific emphasis on symptoms at end of life, and modes of death. Mode of death was described in 2 studies and was predominantly through respiratory or heart failure.
Yonas et al15 found that heart failure was associated with hospitalization (HR 2.37 [1.48, 3.79; p < 0.001], high heterogeneity [I2, 82%; p < 0.001]) and poor outcome (OR of 2.86 [2.07, 3.95; p < 0.001] high heterogeneity [I2, 80%; p < 0.001]) in COVID-19. Preexisting heart failure was associated with higher mortality (OR of 3.46 [2.52, 4.75; p < 0.001] moderately high heterogeneity [I2, 77%; p < 0.001]).
Several studies investigated cardiac and inflammatory biomarkers as prognostic of severe outcomes10, 13, 14. Dalia et al10 state that cardiac involvement in COVID-19 infection appears to significantly adversely impact patient prognosis and survival. Pre-existence of CHF, and high cardiac biomarkers such as NT-pro BNP and CK-MB levels in COVID-19 patients correlates with worse outcomes. Shoar et al14 states that severe clinical presentation and elevated biomarkers in COVID-19 patients with established risk factors can predict mortality from cardiovascular causes.
In a cohort study published after all of the systematic reviews included in the present summary, Alvarez-Garcia et al17conclude that history of heart failure is associated with higher risk of mechanical ventilation and mortality for COVID-19 hospitalised patients. Left ventricular ejection fraction was not found to be independently associated with outcome.
In another large cohort study cited by the Centers for Disease Control and Prevention (United States), Rosenthal et al18 reviewed the characteristics of patients with COVID-19 treated in US hospitals between 1 of April and 31 May, 2020; and showed that adjusted odds ratio for in hospital mortality for those with CHF was 1.37 (95% CI, 1.26–1.49).
Jarkovsky et al16 developed a prediction model based on elementary independent variables called a COVIDogram that allows the identification with high reliability of patients at risk of severe COVID-19.
A modelling study by O’Hearn et al20estimated absolute and proportional COVID-19 hospitalizations in US adults attributable to 4 major US cardiometabolic conditions, separately and jointly, and by race/ethnicity, age, and sex. As of November 18, 2020, an estimated 906 849 COVID-19 hospitalizations occurred in US adults. Of these, an estimated 20.5% (95% uncertainty interval [UIs], 18.9-22.1) of COVID-19 hospitalizations were attributable to diabetes mellitus, 30.2% (UI, 28.2-32.3) to total obesity (body mass index ≥30 kg/m2), 26.2% (UI, 24.3-28.3) to hypertension, and 11.7% (UI, 9.5-14.1) to heart failure. Considered jointly, 63.5% (UI, 61.6-65.4) or 575 419 (UI, 559 072-593 412) of COVID-19 hospitalizations were attributable to these 4 conditions. The authors conclude that a substantial proportion of US COVID-19 hospitalizations appear attributable to major cardiometabolic conditions.
Clift et al19 developed a living risk prediction algorithm (QCOVID) to estimate hospital admission and mortality outcomes from Coronavirus Disease 2019 in adults. The QCOVID population based risk algorithm performed well, showing very high levels of discrimination for deaths and hospital admissions due to COVID-19. The model can be recalibrated for different time periods and has the potential to be dynamically updated as the pandemic evolves.
Irish and/or International Guidance
| Level 1 |
Heart failure is listed in Table 5.2 under chronic heart and vascular diseases associated with a very high risk or high risk of severe COVID-19 disease.
| Level 1 |
Heart Conditions
Having heart conditions such as heart failure, coronary artery disease, cardiomyopathies and possibly high blood pressure (hypertension) can make you more likely to get severely ill from COVID-19.
| Level 1 |
Risk Factors Associated with In-Hospital Mortality in a US National Sample of Patients with COVID-19 (USA)
CDC cite Rosenthal et al18, a retrospective cohort study of 64,781 patients with COVID-19 which included both inpatients and hospital-based outpatients with laboratory-diagnosed COVID-19 between April 1 and May 31, 2020. The study was designed to examine risk factors associated with in-hospital mortality, and the analysis was conducted using multivariable logistic regression among adults for whom sex was known, adjusting for demographic characteristics, visit characteristics, comorbidities, and hospital characteristics, among other factors.
Main Findings
- 29.9% of inpatients and 74.9% of outpatients diagnosed with COVID-19 had no comorbidities as defined in the study.
- The most common comorbidities were hypertension (30,236 [46.7%]), hyperlipidemia (18,744 [28.9%]), diabetes (18,091 [27.9%]), and chronic pulmonary disease (10,434 [16.1%]).
- 6,849 (19.4%) inpatients were admitted to an ICU.
- 5,628 (15.9%) inpatients received invasive mechanical ventilation.
- 7,355 (20.3%) inpatients died in the hospital.
- Very old age was the risk factor most strongly associated with death (eg age 80 years vs. 18–34 years: odds ratio [OR], 16.20; 95% confidence interval [CI], 11.58–22.67; P <0.001).
- Preexisting comorbidities significantly associated with increased odds of in-hospital mortality included congestive heart failure, 37% increase (aOR=1.37; 95% CI, 1.26–1.49).
- The risk for inpatient mortality was higher with increasing number of comorbid conditions.
| Level 1 |
Among 378,048 death certificates with the ICD-10 code U07.1 [COVID-19], 94.5% (357,133) had at least one other ICD-10 code, whereas 5.5% (20,915) listed only U07.1; 330,198 (87%) listed COVID-19 in Part I. Death certificates with only U07.1 and no other diagnosis accounted for 2.9%–6.6% of death certificates for decedents across all age, sex and racial/ethnic categories.
Overall, 97.3% of 357,133 death certificates with at least one other diagnosis (91.9% of all 378,048 death certificates) were noted to have a co-occurring diagnosis that was a plausible chain-of-event condition (eg pneumonia, respiratory failure, adult respiratory distress syndrome, cardiac arrest, or sepsis), or significant contributing condition (eg hypertension, diabetes, dementia, or chronic obstructive pulmonary disease), or both. The most frequent chain-of-event ICD-10 diagnosis codes on 330,198 death certificates that listed COVID-19 on Part I of the death certificate were J18.9 (pneumonia) (45%) and J96.0 (acute respiratory failure) (20%); the most frequent significant contributing condition ICD-10 codes were I10 (essential hypertension) (18%) and E14.9 (diabetes mellitus) (10%). Nearly 75% of all death certificates had a chain-of-event condition, alone or in combination with a significant contributing condition; this finding was noted for adults aged 18–84 years, males and females, persons of all races and ethnicities, those who died in inpatient and outpatient or emergency department settings, and those whose manner of death was listed as natural.
Nearly 18% of death certificates had a co-occurring significant contributing condition. This finding was more frequent for death certificates indicating that the death occurred in the decedent’s home (38.3%), a nursing home or long-term care facility (38.5%), or hospice facility (23.2%).
TABLE 2. Highest-frequency International Classification of Diseases, Tenth Revision (ICD-10) codes listed in death certificates with COVID-19 in Part I of death certificate and at least one diagnosis other than COVID-19 (330,198) — National Center for Health Statistics, United States, January–December 2020
Condition (ICD-10 code)
Conditions listed as chain-of-event conditions on ≥1% of death certificates
Pneumonia, unspecified (J18.9)
Acute respiratory failure (J96.0)
Respiratory failure, unspecified (J96.9)
Cardiac arrest, unspecified (I46.9)
Adult respiratory distress syndrome (J80)
Sepsis, unspecified (A41.9)
Viral pneumonia, unspecified (J12.9)
Asphyxia (R09.0)
Respiratory arrest (R09.2)
Conditions listed as significant contributing conditions on ≥1% of death certificates§
Essential (primary) hypertension (I10)
Unspecified diabetes mellitus without complications (E14.9)
Unspecified dementia (F03)
Chronic obstructive pulmonary disease, unspecified (J44.9)
Atherosclerotic heart disease (I25.1)
Type 2 diabetes mellitus without complications (E11.9)
Atrial fibrillation and flutter (I48)
Congestive heart failure (I50.0)
Tobacco use (F17.9)
Chronic kidney disease, unspecified (N18.9)
Alzheimer disease, unspecified (G30.9)
Hypertensive heart disease without (congestive) heart failure (I11.9)
Hyperlipidemia, unspecified (E78.5)
Other specified disorders of kidney and ureter (N28.8)
Obesity, unspecified (E66.9)
Chronic kidney disease, stage 5 (N18.5)
Stroke, not specified as hemorrhage or infarction (I64)
Heart failure, unspecified (I50.9)
§ COVID-19 diagnosis listed in Part I of the death certificate; significant contributing conditions listed below the COVID-19 line in Part I or in Part II of the death certificate.
| Level 1 |
Main Points
- Cardiovascular involvement is common in patients with severe COVID-19 disease.
- Cardiomyopathy with SARS-CoV-2 may result from myocarditis, profound systemic inflammation, and/or microvascular dysfunction.
- Evidence of cardiac involvement is associated with worse prognosis in COVID-19.
- Cardiac diagnostic imaging or laboratory testing may be useful in certain patients, but risk vs. benefit and whether additional testing will result in change in management should be considered.
- Background guideline-directed therapy is important to be continued in ambulatory patients who are hemodynamically stable and do not have contraindications.
- Experimental strategies including antiviral, anti-inflammatory, convalescent sera are being considered as part of research protocols or compassionate use for COVID-19 infection with or without cardiac involvement.
- Those with underlying cardiovascular disease are particularly susceptible to cardiovascular complications and mortality with COVID-19.
| Level 1 |
See Section: RISK FACTORS FOR HOSPITALIZATION DUE TO COVID-19
Overall, age, comorbidities and severity of disease were found to be strong predictors of hospital admission. Heart failure is among the factors reported to have an especially high risk.
See Section: RISK FACTORS FOR SEVERE ILLNESS, INCLUDING ICU ADMISSION, DUE TO COVID-19
Overall, increasing age, male sex, morbid obesity, heart failure, diabetes and unknown smoking status were associated with increased risk for severe illness.
| Level 1 |
Epidemiological data from China show that 20% or more of COVID‐19 patients have concomitant cardiovascular disease and these patients are more likely to develop life-threatening complications from the infection. The risk of complications may be higher in patients with heart failure not only because they are older and have more comorbidities, but also due to the specific characteristics of this syndrome. It has also been suggested that COVID‐19 may cause or precipitate myocardial injury and worsen heart failure due to a cytokine storm‐related hyper‐inflammation syndrome.
To date, most studies have only reported the prevalence of cardiovascular disease, without details about the prevalence of heart failure. The prevalence of heart failure was 4% in the study by Shi et al, with similar percentages for cardiomyopathy in the study by Guo et al. These values are likely higher in the COVID‐19 patients in Europe or the United States because of their older age. In the study by Zhou et al from the Wuhan area in China, including 191 COVID‐19 inpatients, 23% of the patients developed heart failure, and the prevalence of heart failure in those who died was markedly higher (52%) than that among those who survived (12%). Similar data were reported in other series.
Evidence Synopsis Resources
| Level 2 |
UpToDate (2021) COVID-19: Cardiac Manifestations in Adults 8
See Section: HEART FAILURE
General Prevalence
Heart Failure (HF) in patients with COVID-19 may be precipitated by acute illness with preexisting known or undiagnosed heart disease, acute hemodynamic stress, or acute myocardial injury. Cardiovascular risk factors and cardiovascular disease are highly prevalent in hospitalized patients with COVID-19. Patients with a known history of HF may suffer an acute decompensation due to the development of COVID-19 disease.
A study of 6439 patients hospitalized with COVID-19 at a hospital in New York found that a history of HF was associated with adverse outcomes, including longer length of stay (8 vs. 6 days), increased risk of mechanical ventilation (22.8% vs. 11.9%; adjusted odds ratio [OR] 3.64, 95% CI 2.56-5.16), and mortality (40.0% vs. 24.9%; adjusted OR 1.88, 95% CI 1.27-2.78). Outcomes among patients with different types of HF were similar, regardless of LV ejection fraction (LVEF).
Limited data are available on the incidence of HF in patients with COVID-19. In a retrospective study of 799 patients hospitalized with COVID-19 in Wuhan, HF was identified as a complication in 49 percent of patients who died and in 3 percent of patients who recovered, despite a less than 1 percent baseline prevalence of chronic HF in the combined groups. In a study of 191 patients hospitalized in two other medical centers in Wuhan, HF was identified in 52 percent of patients who died and in 12 percent of patients who recovered. These studies do not provide sufficient information to determine the incidence of new-onset HF with COVID-19, as there was not enough information provided regarding prior history of HF and risk factors for HF.
Although acute HF incidence was not documented in some series of hospitalized patients with COVID-19, elevated natriuretic peptides (such as B-type natriuretic peptide [BNP] and N-terminal pro-BNP [NT-proBNP]) are common, particularly in patients with evidence of cardiac injury.
Right Heart Failure
Acute cor pulmonale (right HF due to acute pulmonary hypertension) precipitated by acute pulmonary embolism or adult respiratory distress syndrome (ARDS) has been described in patients with COVID-19. Patients with COVID-19 are at risk for development of ARDS. Venous thromboembolism (including extensive deep vein thrombosis and pulmonary embolism) is common in acutely ill patients with COVID-19.
Cardiogenic Shock
Case reports have described patients with COVID-19 and acute onset of cardiogenic shock treated with inotrope and mechanical circulatory support and, in some cases, venoarterial extracorporeal membrane oxygenation (VA-ECMO). Rapid recovery within several days has been described in several reported cases with a time course suggestive of possible stress cardiomyopathy. Although fulminant myocarditis was suspected in some cases of cardiogenic shock with recovery of ventricular function over days or weeks, this diagnosis has generally not been established, as endomyocardial biopsy was either not performed or, when performed, did not show findings of myocarditis.
Multisystem Inflammatory Syndrome in Adults (MIS-A)
Multisystem inflammatory syndrome (MIS) was initially described in children (MIS-C) with recent COVID-19 infection as an illness similar to Kawasaki disease associated with fever, gastrointestinal symptoms, shock, LV systolic dysfunction, and elevated inflammatory markers.
Similar cases of MIS have been described in young to middle-aged adults (MIS-A) also presenting with fever, gastrointestinal symptoms, and shock with vasoplegia, LV systolic dysfunction, and elevated inflammatory markers. Many of these patients had history of recent COVID-19 and had positive SARS-CoV-2 antibody tests, with fewer having positive SARS-CoV-2 reverse transcription polymerase chain reaction tests. This diagnosis should be considered in young adults presenting in inflammatory shock. This syndrome appears to be highly responsive to parenteral steroids.
Irish and/or International Literature
| Level 1 |
Background: Understanding the factors associated with disease severity and mortality in Coronavirus disease 2019 (COVID-19) is imperative to effectively triage patients. The authors performed a systematic review to determine the demographic, clinical, laboratory and radiological factors associated with severity and mortality in COVID-19.
Methods: A literature search of PubMed, Embase and WHO database for English language articles from inception until May 8, 2020. The authors included observational studies with direct comparison of clinical characteristics between: a. patients who died and those who survived; or b. patients with severe disease and those without severe disease. Data extraction and quality assessment were performed by two authors independently.
Results: Among 15680 articles from the literature search, 109 articles were included in the analysis. The risk of mortality was higher in patients with increasing age, male gender (RR 1.45, 95%CI 1.23-1.71), dyspnea (RR 2.55, 95%CI 1.88-2.46), diabetes (RR 1.59, 95%CI 1.41-1.78), hypertension (RR 1.90, 95%CI 1.69-2.15). Congestive heart failure (OR 4.76, 95%CI 1.34-16.97), hilar lymphadenopathy (OR 8.34, 95%CI 2.57-27.08), bilateral lung involvement (OR 4.86, 95%CI 3.19-7.39) and reticular pattern (OR 5.54, 95%CI 1.24-24.67) were associated with severe disease. Clinically relevant cut-offs for leucocytosis (>10.0 x109/L), lymphopenia (< 1.1 x109/L), elevated C-reactive protein(>100mg/L), LDH(>250U/L) and D-dimer(>1mg/L) had higher odds of severe disease and greater risk of mortality.
Conclusion: Knowledge of the factors associated of disease severity and mortality identified in our study may assist in clinical decision-making and critical-care resource allocation for patients with COVID-19.
| Level 1 |
Background: Coronavirus disease 2019 (COVID-19) has been reported to cause worse outcomes in patients with underlying cardiovascular disease, especially in patients with acute cardiac injury, which is determined by elevated levels of high-sensitivity troponin. There is a paucity of data on the impact of congestive heart failure (CHF) on outcomes in COVID-19 patients.
Methods: A literature search of PubMed/Medline, EMBASE, and Google Scholar databases from 11/1/2019 until 06/07/2020, and identified all relevant studies reporting cardiovascular comorbidities, cardiac biomarkers, disease severity, and survival. Pooled data from the selected studies was used for meta-analysis to identify the impact of risk factors and cardiac biomarker elevation on disease severity and/or mortality.
Results: The authors collected pooled data on 5967 COVID-19 patients from 20 individual studies and found that both non-survivors and those with severe disease had an increased risk of acute cardiac injury and cardiac arrhythmias, our pooled relative risk (RR) was – 8.52 (95% CI 3.63-19.98) (p < 0.001); and 3.61 (95% CI 2.03-6.43) (p = 0.001), respectively. Mean difference in the levels of Troponin-I, CK-MB, and NT-proBNP was higher in deceased and severely infected patients. The RR of in-hospital mortality was 2.35 (95% CI 1.18-4.70) (p = 0.022) and 1.52 (95% CI 1.12-2.05) (p = 0.008) among patients who had pre-existing CHF and hypertension, respectively.
Conclusion: Cardiac involvement in COVID-19 infection appears to significantly adversely impact patient prognosis and survival. Pre-existence of CHF, and high cardiac biomarkers such as NT-pro BNP and CK-MB levels in COVID-19 patients correlates with worse outcomes.
Figure: Risk ratio of death for patients with CHF compared to those without CHF

| Level 1 |
Background: High rates of cardiovascular disease (CVD) have been reported among patients with novel coronavirus disease (COVID-19). Meanwhile there have been disparities among different studies about CVD burden in COVID-19 patients. The authors aimed to study CVD burden among COVID-19 patients using a systematic review and meta-analysis.
Methods: A systematic literature search including PubMed, Embase, Cochrane Library, Scopus, Web of Science as well as the medRxiv pre-print database. Hand searches were also conducted in journal websites and Google Scholar. Meta-analyses were carried out for Odds Ratio (OR) of mortality and Intensive Care Unit (ICU) admission for different CVDs. A descriptive meta-analysis on different CVDs was also carried out.
Results: 56 studies were included in the meta-analysis for ICU admission and mortality outcome, and 198 studies in the meta- analysis for descriptive outcomes, comprising 159,698 COVID-19 patients. The results of the meta-analysis indicated that acute cardiac injury, (OR: 13.29, 95% CI 7.35-24.03), hypertension (OR: 2.60, 95% CI 2.11-3.19), heart failure (OR: 6.72, 95% CI 3.34-13.52), arrhythmia (OR: 2.75, 95% CI 1.43-5.25), coronary artery disease (OR: 3.78, 95% CI 2.42-5.90) and cardiovascular disease (OR: 2.61, 95% CI 1.89-3.62) were significantly associated with mortality. Arrhythmia (OR: 7.03, 95% CI 2.79-17.69), acute cardiac injury (OR: 15.58, 95% CI 5.15-47.12), coronary heart disease (OR: 2.61, 95% CI 1.09-6.26), cardiovascular disease (OR: 3.11, 95% CI 1.59-6.09) and hypertension (OR: 1.95, 95% CI 1.41-2.68) were also significantly associated with ICU admission in COVID-19 patients.
Conclusion: These findings revealed a high burden of CVDs among COVID-19 patients which was significantly associated with mortality and ICU admission. Proper management of CVD patients with COVID-19 and monitoring COVID-19 patients for acute cardiac conditions is highly recommended.
Figure: Prevalence of cardiovascular complications among COVID-19 patients

| Level 1 |
Methods: The authors conducted a systematic review of published evidence for symptoms in patients with COVID-19 with a specific emphasis on symptoms at end of life and on modes of death. Inclusion: Prospective or retrospective studies detailing symptom presence and/or cause or mode of death from COVID-19.
Results: 12 studies met the inclusion criteria and gave details of symptom burden: 4 of these specifically in the dying, and 2 detailing cause or mode of death. Cough, breathlessness, fatigue and myalgia are significant symptoms in people hospitalised with COVID-19. Dyspnoea is the most significant symptom in the dying. The mode of death was described in 2 studies and is predominantly through respiratory or heart failure.
Conclusions: There remains a dearth of information regarding symptom burden and mode of death to inform decisions regarding end-of-life care in patients dying with COVID-19. Rapid data gathering on the mode of death and the profile of symptoms in the dying and their prevalence and severity in areas where COVID-19 is prevalent will provide important intelligence for clinicians.
| Level 1 |
The coronavirus disease 2019 (COVID-19) pandemic has drastically affected global health. Despite several studies, there is still a dearth of data regarding the mechanisms of cardiac injury, clinical presentation, risk factors, and treatment of COVID-19-associated cardiovascular disease. This systematic review and meta-analysis aims to define the clinical, electrocardiographic and pathologic spectrum of cardiovascular disease (CVD), frequency of elevated cardiac and inflammatory biomarkers and their relationship with severity of the disease, and mortality in COVID-19 patients; and to develop a triage risk stratification tool (TRST) that can serve as a guide for the timely recognition of high-risk patients and mechanism-targeted therapy.
METHODS: A literature search of PubMed and Embase to identify relevant studies. Data selection was in concordance with PRISMA guidelines. Results were presented as pooled frequencies, odds ratios, standardized mean differences (SMDs), and forest and funnel plots.
Results: A total of 54 studies were gathered, of which 35 were included in the meta-analysis. Acute cardiac injury occurred in more than 25% of cases; mortality was 20 times higher; and admission to intensive care unit was 13.5 times higher. Hypertension was the most common pre-existing comorbidity with a frequency of 29.2%, followed by diabetes mellitus (13.5%). The deceased group of patients had higher cardiac and inflammatory biomarkers, with statistically significant SMD, compared with survivors. Pediatric patients were predominantly mildly affected. However, despite the lower frequently, the presentation was very similar to Kawasaki disease or Kawasaki shock syndrome. This latter presentation has been called multisystem inflammatory syndrome in children (MIS-C).
Conclusions: There is a wide spectrum of cardiac involvement in COVID-19 patients, and hence a Triage Risk Stratification Tool can serve as a guide for the timely recognition of high-risk patients and mechanism-targeted therapy.
| Level 1 |
Since the emergence of COVID-19, a number of studies have reported the presence of cardiovascular diseases in affected patients and linked these diseases with a higher risk of mortality. The authors conducted a literature search in Medline/PubMed to identify original cohorts comparing data between survivors and non-survivors from COVID-19. The presence of cardiovascular events and related biomarkers were compared between the 2 groups. Data on 1,845 hospitalized patients with COVID-19 were pooled from 12 comparative studies. The overall mortality rate in relation to COVID-19 was 17.6%. Men aged > 50 years were more likely to die from COVID-19. Significant co-morbidities contributing to mortality were hypertension, diabetes mellitus, smoking, a previous history of cardiovascular disease including chronic heart failure, and cerebrovascular accidents. A significant relationship was observed between mortality and patient presentation with dyspnea, fatigue, tachycardia, and hypoxemia. Cardiovascular disease-related laboratory biomarkers related to mortality were elevated serum level of lactate dehydrogenase, creatine kinase, brain natriuretic peptide, and cardiac troponin I. Adverse cardiovascular disease-related clinical events preceding death were shock, arrhythmias, and acute myocardial injury. Severe clinical presentation and elevated biomarkers in COVID-19 patients with established risk factors can predict mortality from cardiovascular causes.
| Level 1 |
Background: Several comorbidities have been associated with an increased risk of severity and mortality in coronavirus disease 2019 (COVID-19), including hypertension, diabetes, cerebrovascular disease, chronic kidney disease, and chronic obstructive pulmonary disease.
Purpose: In this systematic review and meta-analysis, the authors attempted to investigate the association between heart failure (HF) and poor outcome in patients with COVID-19.
Methods: A systematic literature search of PubMed, EuropePMC, SCOPUS, Cochrane Central Database and medRxiv with the search terms “heart failure” and “COVID-19.” The outcome of interest was mortality and poor prognosis ⎯ defined by incidence of severe COVID-19 infection, admission to ICU, and use of ventilator ⎯ in patients with preexisting heart failure with coronavirus disease.
Results: The authors identified 92 potentially relevant articles. 74 studies were excluded due to the following reasons: 4 studies were systematic reviews, 2 studies were meta-analyses, 3 articles were literature reviews, and 65 articles did not report on the outcome of interest. The remaining 18 studies were included in the qualitative synthesis and meta-analysis. There were 21,640 patients from 18 studies. HF was associated with hospitalization in COVID-19 (HR 2.37 [1.48, 3.79; p < 0.001], high heterogeneity [I2, 82%; p < 0.001]). HF was associated with a poor outcome (OR of 2.86 [2.07, 3.95; p < 0.001] high heterogeneity [I2, 80%; p < 0.001]). Preexisting HF was associated with higher mortality (OR of 3.46 [2.52, 4.75; p < 0.001] moderately high heterogeneity [I2, 77%; p < 0.001]).
Conclusion: Patients with heart failure are at increased risk for hospitalization, poor outcome and death from COVID-19. A significant difference in mortality between patients with and without heart failure was observed, patients with heart failure having a higher mortality.
Figure: Risk of hospitalization in patients with pre-existing heart failure

Figure: Meta-analysis: heart failure and poor outcome

Figure: Meta-analysis: pooled HR of mortality in COVID-19 patients with pre-existing heart failure

Figure: Meta-analysis: mortality in COVID-19 patients with pre-existing heart failure compared to non-HF patients

Figure: Meta-analysis: incidence of new-onset heart failure in COVID-19 patients
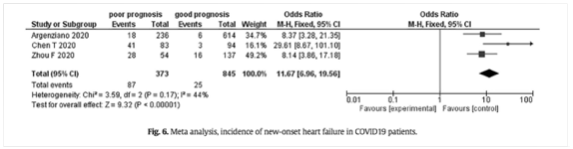
| Level 4 |
Objectives: COVID-19 might either be entirely asymptomatic or manifest itself with a large variability of disease severity. It is beneficial to identify early patients with a high risk of severe course. The aim of the analysis was to develop a prognostic model for the prediction of the severe course of acute respiratory infection.
Design: A population-based study.
Setting: Czech Republic.
Participants: The first 7455 consecutive patients with COVID-19 who were identified by reverse transcription-PCR testing from 1 March 2020 to 17 May 2020.
Primary outcome: Severe course of COVID-19.
Result: 6.2% of patients developed a severe course of COVID-19. Age, male sex, chronic kidney disease, chronic obstructive pulmonary disease, recent history of cancer, chronic heart failure, acid-related disorders treated with proton-pump inhibitors and diabetes mellitus were found to be independent negative prognostic factors (Area under the ROC Curve (AUC) was 0.893). The results were visualised by risk heat maps, a diagram subsequently called a COVIDogram. Acid-related disorders treated with proton-pump inhibitors might represent a negative prognostic factor.
Conclusion: The authors developed a very simple prediction model called a COVIDogram, which is based on elementary independent variables (age, male sex and the presence of several chronic diseases) and represents a tool that makes it possible to identify with high reliability patients who are at risk of a severe course of COVID-19. Obtained results open clinically relevant question about the role of acid-related disorders treated by proton-pump inhibitors as predictor for severe course of COVID-19.
| Level 4 |
Background: Patients with pre-existing heart failure (HF) are likely at higher risk for adverse outcomes in coronavirus disease-2019 (COVID-19), but data on this population are sparse.
Objectives: This study described the clinical profile and associated outcomes among patients with HF hospitalized with COVID-19.
Methods: A retrospective analysis of 6,439 patients admitted for COVID-19 at 1 of 5 Mount Sinai Health System hospitals in New York City between February 27 and June 26, 2020. Clinical characteristics and outcomes (length of stay, need for intensive care unit, mechanical ventilation, and in-hospital mortality) were captured from electronic health records. For patients identified as having a history of HF by International Classification of Diseases-9th and/or 10th Revisions codes, manual chart abstraction informed etiology, functional class, and left ventricular ejection fraction (LVEF).
Results: Mean age was 63.5 years, and 45% were women. Compared with patients without HF, those with previous HF experienced longer length of stay (8 days vs. 6 days; p < 0.001), increased risk of mechanical ventilation (22.8% vs. 11.9%; adjusted odds ratio: 3.64; 95% confidence interval: 2.56 to 5.16; p < 0.001), and mortality (40.0% vs. 24.9%; adjusted odds ratio: 1.88; 95% confidence interval: 1.27 to 2.78; p = 0.002). Outcomes among patients with HF were similar, regardless of LVEF or renin-angiotensin-aldosterone inhibitor use.
Conclusions: History of HF was associated with higher risk of mechanical ventilation and mortality among patients hospitalized for COVID-19, regardless of LVEF.
| Level 4 |
Importance: Coronavirus disease 2019 (COVID-19) has infected more than 8.1 million US residents and killed more than 221 000. There is a dearth of research on epidemiology and clinical outcomes in US patients with COVID-19.
Objectives: To characterize patients with COVID-19 treated in US hospitals and to examine risk factors associated with in-hospital mortality.
Design, setting, and participants: This cohort study was conducted using Premier Healthcare Database, a large geographically diverse all-payer hospital administrative database including 592 acute care hospitals in the United States. Inpatient and hospital-based outpatient visits with a principal or secondary discharge diagnosis of COVID-19 (International Classification of Diseases, Tenth Revision, Clinical Modification diagnosis code, U07.1) between April 1 and May 31, 2020 were included.
Exposures: Characteristics of patients were reported by inpatient/outpatient and survival status. Risk factors associated with death examined included patient characteristics, acute complications, comorbidities, and medications.
Main outcomes and measures: In-hospital mortality, intensive care unit (ICU) admission, use of invasive mechanical ventilation, total hospital length of stay (LOS), ICU LOS, acute complications, and treatment patterns.
Results: Overall, 64 781 patients with COVID-19 (29 479 [45.5%] outpatients; 35 302 [54.5%] inpatients) were analyzed. The median (interquartile range [IQR]) age was 46 (33-59) years for outpatients and 65 (52-77) years for inpatients; 31 968 (49.3%) were men, 25 841 (39.9%) were White US residents, and 14 340 (22.1%) were Black US residents. In-hospital mortality was 20.3% among inpatients (7164 patients). A total of 5625 inpatients (15.9%) received invasive mechanical ventilation, and 6849 (19.4%) were admitted to the ICU. Median (IQR) inpatient LOS was 6 (3-10) days. Median (IQR) ICU LOS was 5 (2-10) days. Common acute complications among inpatients included acute respiratory failure (19 706 [55.8%]), acute kidney failure (11 971 [33.9%]), and sepsis (11 910 [33.7%]). Older age was the risk factor most strongly associated with death (eg age ≥80 years vs. 18-34 years: odds ratio [OR], 16.20; 95% CI, 11.58-22.67; P < .001). Receipt of statins (OR, 0.60; 95% CI, 0.56-0.65; P < .001), angiotensin-converting enzyme inhibitors (OR, 0.53; 95% CI, 0.46-0.60; P < .001) and calcium channel blockers (OR, 0.73; 95% CI, 0.68-0.79; P < .001) was associated with decreased odds of death. Compared with patients with no hydroxychloroquine or azithromycin, patients with both azithromycin and hydroxychloroquine had increased odds of death (OR, 1.21; 95% CI, 1.11-1.31; P < .001).
Conclusions and relevance: In this cohort study of patients with COVID-19 infection in US acute care hospitals, COVID-19 was associated with high ICU admission and in-hospital mortality rates. Use of statins, angiotensin-converting enzyme inhibitors, and calcium channel blockers were associated with decreased odds of death. Understanding the potential benefits of unproven treatments will require future randomized trials.
| Level 4 |
Objective: To derive and validate a risk prediction algorithm to estimate hospital admission and mortality outcomes from coronavirus disease 2019 (COVID-19) in adults.
Design: Population based cohort study.
Setting and participants: QResearch database, comprising 1205 general practices in England with linkage to COVID-19 test results, Hospital Episode Statistics, and death registry data. 6.08 million adults aged 19-100 years were included in the derivation dataset and 2.17 million in the validation dataset. The derivation and first validation cohort period was 24 January 2020 to 30 April 2020. The second temporal validation cohort covered the period 1 May 2020 to 30 June 2020.
Main outcome measures: The primary outcome was time to death from COVID-19, defined as death due to confirmed or suspected COVID-19 as per the death certification or death occurring in a person with confirmed severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection in the period 24 January to 30 April 2020. The secondary outcome was time to hospital admission with confirmed SARS-CoV-2 infection. Models were fitted in the derivation cohort to derive risk equations using a range of predictor variables. Performance, including measures of discrimination and calibration, was evaluated in each validation time period.
Results: 4384 deaths from COVID-19 occurred in the derivation cohort during follow-up and 1722 in the first validation cohort period and 621 in the second validation cohort period. The final risk algorithms included age, ethnicity, deprivation, body mass index, and a range of comorbidities. The algorithm had good calibration in the first validation cohort. For deaths from COVID-19 in men, it explained 73.1% (95% confidence interval 71.9% to 74.3%) of the variation in time to death (R2); the D statistic was 3.37 (95% confidence interval 3.27 to 3.47), and Harrell’s C was 0.928 (0.919 to 0.938). Similar results were obtained for women, for both outcomes, and in both time periods. In the top 5% of patients with the highest predicted risks of death, the sensitivity for identifying deaths within 97 days was 75.7%. People in the top 20% of predicted risk of death accounted for 94% of all deaths from COVID-19.
Conclusion: The QCOVID population based risk algorithm performed well, showing very high levels of discrimination for deaths and hospital admissions due to COVID-19. The absolute risks presented, however, will change over time in line with the prevailing SARS-C0V-2 infection rate and the extent of social distancing measures in place, so they should be interpreted with caution. The model can be recalibrated for different time periods, however, and has the potential to be dynamically updated as the pandemic evolves.
| Level 6 |
BACKGROUND: Risk of coronavirus disease 2019 (COVID-19) hospitalization is robustly linked to cardiometabolic health. The authors estimated the absolute and proportional COVID-19 hospitalizations in US adults attributable to 4 major US cardiometabolic conditions, separately and jointly, and by race/ethnicity, age, and sex.
METHODS AND RESULTS: The authors used the best available estimates of independent associations of cardiometabolic conditions with a risk of COVID-19 hospitalization; nationally representative data on cardiometabolic conditions from the National Health and Nutrition Examination Survey 2015 to 2018; and US COVID-19 hospitalizations stratified by age, sex, and race/ethnicity from the Centers for Disease Control and Prevention’s Coronavirus Disease 2019-Associated Hospitalization Surveillance Network database and from the COVID Tracking Project to estimate the numbers and proportions of COVID-19 hospitalizations attributable to diabetes mellitus, obesity, hypertension, and heart failure. Inputs were combined in a comparative risk assessment framework, with probabilistic sensitivity analyses and 1000 Monte Carlo simulations to jointly incorporate stratum-specific uncertainties in data inputs. As of November 18, 2020, an estimated 906 849 COVID-19 hospitalizations occurred in US adults. Of these, an estimated 20.5% (95% uncertainty interval [UIs], 18.9-22.1) of COVID-19 hospitalizations were attributable to diabetes mellitus, 30.2% (UI, 28.2-32.3) to total obesity (body mass index ≥30 kg/m2), 26.2% (UI, 24.3-28.3) to hypertension, and 11.7% (UI, 9.5-14.1) to heart failure. Considered jointly, 63.5% (UI, 61.6-65.4) or 575 419 (UI, 559 072-593 412) of COVID-19 hospitalizations were attributable to these 4 conditions. Large differences were seen in proportions of cardiometabolic risk-attributable COVID-19 hospitalizations by age and race/ethnicity, with smaller differences by sex.
CONCLUSIONS: A substantial proportion of US COVID-19 hospitalizations appear attributable to major cardiometabolic conditions. These results can help inform public health prevention strategies to reduce COVID-19 healthcare burdens.
| Level 7 |
Williamson EJ et al (2020) Factors associated with COVID-19-related death using OpenSAFELY 21
Working on behalf of NHS England, the authors created OpenSAFELY ⎯ a secure health analytics platform that covers 40% of all patients in England and holds patient data within the existing data centre of a major vendor of primary care electronic health records ⎯ and used OpenSAFELY to examine factors associated with COVID-19-related death. Primary care records of 17,278,392 adults were pseudonymously linked to 10,926 COVID-19-related deaths. COVID-19-related death was associated with: being male (hazard ratio (HR) 1.59 (95% CI 1.53-1.65)); greater age and deprivation (both with a strong gradient); diabetes; severe asthma; and various other medical conditions. Compared with people of White ethnicity, Black and South Asian people were at higher risk, even after adjustment for other factors (HR 1.48 (1.29-1.69) and 1.45 (1.32-1.58), respectively).
| Level 7 |
Long B et al (2020) Cardiovascular complications in COVID-1922
Background: The coronavirus disease of 2019 (COVID-19) is caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). While systemic inflammation and pulmonary complications can result in significant morbidity and mortality, cardiovascular complications may also occur.
Objective: This brief report evaluates cardiovascular complications in the setting of COVID-19 infection.
Discussion: The current COVID-19 pandemic has resulted in over one million infected worldwide and thousands of death. The virus binds and enters through angiotensin-converting enzyme 2 (ACE2). COVID-19 can result in systemic inflammation, multi-organ dysfunction, and critical illness. The cardiovascular system is also affected, with complications including myocardial injury, myocarditis, acute myocardial infarction, heart failure, dysrhythmias, and venous thromboembolic events. Current therapies for COVID-19 may interact with cardiovascular medications.
Conclusions: Emergency clinicians should be aware of these cardiovascular complications when evaluating and managing the patient with COVID-19.
References
1.Health Service Executive. National Immunization Advisory Committee (2021) Immunisation Guidelines. Chapter 5a: COVID-19. https://www.hse.ie/eng/health/immunisation/hcpinfo/guidelines/covid19.pdf. Accessed 27 May 2021.
2. Centers for Disease Control and Prevention (United States) (2021) Certain Medical Conditions and Risk for Severe COVID-19. https://www.cdc.gov/coronavirus/ Accessed 27 May 2021.2019-ncov/need-extra-precautions/people-with-medical-conditions.html. Accessed 27 May 2021.
3. Centers for Disease Control and Prevention (United States) (2021) Underlying Medical Conditions Associated with High Risk for Severe COVID-19: Information for Healthcare Providers. https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-care/underlyingconditions.html. Accessed 27 May 2021.
4. Centers for Disease Control and Prevention (United States) (2021) Death Certificate-Based ICD-10 Diagnosis Codes for COVID-19 Mortality Surveillance — United States, January-December 2020. https://www.cdc.gov/mmwr/volumes/70/wr/mm7014e2.htm?s_cid=mm7014e2_x. Accessed 27 May 2021.
5. American College of Cardiology (2020) Management of the hospitalized COVID-19 patient with acute cardiomyopathy or heart failure. https://www.acc.org/latest-in-cardiology/articles/2020/04/16/14/42/management-of-the-hospitalized-covid-19-coronavirus-2019-patient-with-acute-cardiomyopathy-or-heart-failure. Accessed 27 May 2021.
6. Norwegian Institute of Public Health (2020) [Rapid Evidence Summary] COVID-19 and risk factors for hospital admission, severe disease and death. https://www.fhi.no/globalassets/dokumenterfiler/rapporter/2020/covid-19-and-risk-factors-for-hospital-admission-severe-disease-and-death-3rd-update-memo-2020-v2.pdf. Accessed 27 May 2021.
7. Chinese Heart Failure Association and European Society of Cardiology (2020) Management of heart failure in patients with COVID-19: a joint position paper. https://onlinelibrary.wiley.com/doi/10.1002/ejhf.1915. Accessed 27 May 2021.
8. UpToDate (2021) COVID-19: Cardiac Manifestations in Adults. https://www.uptodate.com/contents/covid-19-cardiac-manifestations-in-adults#:~:text=Adults%20with%20COVID%2D19%20present,cardiac%20arrhythmias%2C%20or%20abnormalities%20on. Accessed 27 May 2021.
9. Chidambaram V, Tun NL, Haque WZ, Majella MG, Sivakumar RK, Kumar A, Hsu AT, Ishak IA, Nur AA, Ayeh SK, Salia EL, Zil-E-Ali A, Saeed MA, Sarena APB, Seth B, Ahmadzada M, Haque EF, Neupane P, Wang KH, Pu TM, Ali SMH, Arshad MA, Wang L, Baksh S, Karakousis PC, Galiatsatos P. Factors associated with disease severity and mortality among patients with COVID-19: A systematic review and meta-analysis. PLoS One. 2020 Nov 18;15(11):e0241541. doi: 10.1371/journal.pone.0241541. PMID: 33206661; PMCID: PMC7673562.
10. Dalia T, Lahan S, Ranka S, Acharya P, Gautam A, Goyal A, Mastoris I, Sauer A, Shah Z. Impact of congestive heart failure and role of cardiac biomarkers in COVID-19 patients: A systematic review and meta-analysis. Indian Heart J. 2021 Jan-Feb;73(1):91-98. doi: 10.1016/j.ihj.2020.12.002. Epub 2020 Dec 6. PMID: 33714416; PMCID: PMC7719198.
11. Hessami A, Shamshirian A, Heydari K, Pourali F, Alizadeh-Navaei R, Moosazadeh M, Abrotan S, Shojaie L, Sedighi S, Shamshirian D, Rezaei N. Cardiovascular diseases burden in COVID-19: Systematic review and meta-analysis. Am J Emerg Med. 2020 Oct 16:S0735-6757(20)30908-6. doi: 10.1016/j.ajem.2020.10.022. Epub ahead of print. PMID: 33268238; PMCID: PMC7561581.
12. Keeley P, Buchanan D, Carolan C, Pivodic L, Tavabie S, Noble S. Symptom burden and clinical profile of COVID-19 deaths: a rapid systematic review and evidence summary. BMJ Support Palliat Care. 2020 Dec;10(4):381-384. doi: 10.1136/bmjspcare-2020-002368. Epub 2020 May 28. PMID: 32467101.
13. Momtazmanesh S, Shobeiri P, Hanaei S, Mahmoud-Elsayed H, Dalvi B, Malakan Rad E. Cardiovascular disease in COVID-19: a systematic review and meta-analysis of 10,898 patients and proposal of a triage risk stratification tool. Egypt Heart J. 2020 Jul 13;72(1):41. doi: 10.1186/s43044-020-00075-z. PMID: 32661796; PMCID: PMC7356124.
14. Shoar S, Hosseini F, Naderan M, Mehta JL. Meta-analysis of Cardiovascular Events and Related Biomarkers Comparing Survivors Versus Non-survivors in Patients With COVID-19. Am J Cardiol. 2020 Nov 15;135:50-61. doi: 10.1016/j.amjcard.2020.08.044. Epub 2020 Sep 8. PMID: 32916148; PMCID: PMC7477637.
15. Yonas E, Alwi I, Pranata R, Huang I, Lim MA, Gutierrez EJ, Yamin M, Siswanto BB, Virani SS. Effect of heart failure on the outcome of COVID-19 – A meta analysis and systematic review. Am J Emerg Med. 2020 Jul 9:S0735-6757(20)30602-1. doi: 10.1016/j.ajem.2020.07.009. Epub ahead of print. PMID: 33071085; PMCID: PMC7347316.
16. Jarkovsky J, Benesova K, Cerny V, Razova J, Kala P, Dolina J, Majek O, Sebestova S, Bezdekova M, Melicharova H, Snajdrova L, Dusek L, Parenica J. COVIDogram as a simple tool for predicting severe course of COVID-19: population-based study. BMJ Open. 2021 Feb 23;11(2):e045442. doi: 10.1136/bmjopen-2020-045442. PMID: 33622955; PMCID: PMC7907625.
17. Alvarez-Garcia J, Lee S, Gupta A, Cagliostro M, Joshi AA, Rivas-Lasarte M, Contreras J, Mitter SS, LaRocca G, Tlachi P, Brunjes D, Glicksberg BS, Levin MA, Nadkarni G, Fayad Z, Fuster V, Mancini D, Lala A. Prognostic Impact of Prior Heart Failure in Patients Hospitalized With COVID-19. J Am Coll Cardiol. 2020 Nov 17;76(20):2334-2348. doi: 10.1016/j.jacc.2020.09.549. Epub 2020 Oct 28. PMID: 33129663; PMCID: PMC7598769.
18. Rosenthal N, Cao Z, Gundrum J, Sianis J, Safo S. Risk Factors Associated With In-Hospital Mortality in a US National Sample of Patients With COVID-19. JAMA Netw Open. 2020 Dec 1;3(12):e2029058. doi: 10.1001/jamanetworkopen.2020.29058. Erratum in: JAMA Netw Open. 2021 Jan 4;4(1):e2036103. PMID: 33301018; PMCID: PMC7729428.
19. Clift AK, Coupland CAC, Keogh RH, Diaz-Ordaz K, Williamson E, Harrison EM, Hayward A, Hemingway H, Horby P, Mehta N, Benger J, Khunti K, Spiegelhalter D, Sheikh A, Valabhji J, Lyons RA, Robson J, Semple MG, Kee F, Johnson P, Jebb S, Williams T, Hippisley-Cox J. Living risk prediction algorithm (QCOVID) for risk of hospital admission and mortality from coronavirus 19 in adults: national derivation and validation cohort study. BMJ. 2020 Oct 20;371:m3731. doi: 10.1136/bmj.m3731. PMID: 33082154; PMCID: PMC7574532.
20. O’Hearn M, Liu J, Cudhea F, Micha R, Mozaffarian D. Coronavirus Disease 2019 Hospitalizations Attributable to Cardiometabolic Conditions in the United States: A Comparative Risk Assessment Analysis. J Am Heart Assoc. 2021 Feb;10(5):e019259. doi: 10.1161/JAHA.120.019259. Epub 2021 Feb 25. Erratum in: J Am Heart Assoc. 2021 Apr 6;10(7):e020858. PMID: 33629868.
21. Williamson EJ, Walker AJ, Bhaskaran K, Bacon S, Bates C, Morton CE, Curtis HJ, Mehrkar A, Evans D, Inglesby P, Cockburn J, McDonald HI, MacKenna B, Tomlinson L, Douglas IJ, Rentsch CT, Mathur R, Wong AYS, Grieve R, Harrison D, Forbes H, Schultze A, Croker R, Parry J, Hester F, Harper S, Perera R, Evans SJW, Smeeth L, Goldacre B. Factors associated with COVID-19-related death using OpenSAFELY. Nature. 2020 Aug;584(7821):430-436. doi: 10.1038/s41586-020-2521-4. Epub 2020 Jul 8. PMID: 32640463.
22. Long B, Brady WJ, Koyfman A, Gottlieb M. Cardiovascular complications in COVID-19. Am J Emerg Med. 2020 Jul;38(7):1504-1507. doi: 10.1016/j.ajem.2020.04.048. Epub 2020 Apr 18. PMID: 32317203; PMCID: PMC7165109.
Produced by the members of the National Health Library and Knowledge Service Evidence Team†. Current as at 24 May 2021. This evidence summary collates the best available evidence at the time of writing and does not replace clinical judgement or guidance. Emerging literature or subsequent developments in respect of COVID-19 may require amendment to the information or sources listed in the document. Although all reasonable care has been taken in the compilation of content, the National Health Library and Knowledge Service Evidence Team makes no representations or warranties expressed or implied as to the accuracy or suitability of the information or sources listed in the document. This evidence summary is the property of the National Health Library and Knowledge Service and subsequent re-use or distribution in whole or in part should include acknowledgement of the service.
Isabelle Delaunois, Medical Librarian, University of Limerick Hospital [Author]; Emma Quinn, Librarian, St. Luke’s General Hospital, Kilkenny [Author, Editor]; Brendan Leen, Area Library Manager, HSE South [Author, Editor]; NIAC Subgroup Contributors: Dr. Peter O’Reilly; Dr. Niamh Bambury; Dr. Geraldine Casey; Dr. Kenneth Beatty; Dr. Paul Mullane; Dr. Philippa White.

This work is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License.
